Arbuscular Mycorrhizal Fungi in the Rhizosphere of Selected Fruit Tree Species in Maiduguri, Nigeria
| Received 31 Oct, 2025 |
Accepted 25 Jan, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Arbuscular Mycorrhizal Fungi (AMF) enhance plant growth, nutrient uptake, and stress tolerance, yet information on the native AMF associated with fruit trees in Maiduguri is limited. This study aimed to investigate the occurrence, diversity, and colonization potential of AMF in the rhizosphere and roots of selected fruit trees to identify dominant species suitable for inoculum production. Materials and Methods: Root and rhizosphere soil samples were collected from Citrus sinensis (Orange), Anacardium occidentale (Cashew), Carica papaya (Pawpaw), Musa sp. (Banana), and Mangifera indica (Mango) in an established orchard at the University of Maiduguri. The AMF spore density, root colonization, and soil physico-chemical properties were determined using standard methods. The AMF morphotypes were identified to genus level, and Pearson correlation was used to assess relationships between soil properties and AMF parameters. Results: The AMF were present in all sampled trees, with root colonization ranging from 49.13% to 69.01%. Fifteen AMF morphotypes representing five genera Glomus, Acaulospora, Funneliformis, Claroideoglomus, and Gigaspora were identified. Glomus was most dominant (40%), followed by Acaulospora (29%), Funneliformis (13%), Claroideoglomus (12%), and Gigaspora (6%). Species richness varied among tree species; pawpaw hosted four of the five genera, while Gigaspora was absent. Soil organic carbon and pH positively influenced AMF spore density and root colonization, whereas bulk density and available phosphorus showed negative correlations. Conclusion: The study identifies dominant native AMF genera in the rhizosphere of fruit trees in Maiduguri, highlighting their potential as inoculum for sustainable fruit production. Soil management targeting pH and organic carbon may enhance AMF symbiosis. Further studies could evaluate the efficacy of these AMF species under field inoculation trials.
| Copyright © 2026 Abdullahi et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Fruit trees are widely cultivated due to their nutritional, medicinal, and economic value, providing vitamins, minerals, antioxidants, and income opportunities through both local and export markets. With the increasing demand for fruits, many people are investing in the establishment of an orchard as a long-term agricultural enterprise. However, successful orchard systems require healthy soils, optimal nutrient supply, and sustainable management practices to ensure productivity over decades. Arbuscular Mycorrhiza Fungi (AMF) form associations with almost 80% of all land plant species and are essential for plants to colonise new areas1,2. Most of these AMF relationships are characterised by a mutual transfer of resources across the mycorrhizal interface and are beneficial to both parties. The AMF receives 4-20% of the photo-synthetically fixed carbon from the host plant in exchange for their positive impact on plant growth and nutrient uptake3. The roots of most plants, including the bulk of tree crops, are colonised by arbuscular mycorrhiza, which are obligate and mutualistic symbionts. To effectively absorb nutrients from the soil and transfer such nutrients to their host plants in return for carbohydrates, arbuscular mycorrhizal fungi create a vast, complex network of hyphae4. They aid in nutrient absorption, especially those that are immobile, like phosphorus, zinc, and copper5.
The AMF can increase resistance or tolerance to abiotic conditions such as drought and heavy metal toxicity, as well as root infections6,7. They play a crucial role in soil aggregation stability and microbial activity, making them essential for sustainable agriculture. In low-input cropping systems, AMF may play a more significant role in preserving soil fertility and in biocontrol of plant diseases than in traditional agriculture8,9. Their distribution in the rhizosphere is influenced by plant species, soil properties, and environmental conditions. Fruit tree species, being perennial crops, develop long-term interactions with AMF, which can play a vital role in sustaining orchard productivity, soil fertility, and resilience against biotic and abiotic stress. Arbuscular mycorrhizal fungi offer several advantages to plants and the environment, depending on the type of plant and the growing conditions and practices10. It could reduce the excessive application of synthetic fertilizer11. Owing to the accrued benefits of mycorrhizal symbiosis, such as improved drought tolerance, which is crucial for fruit trees in semi-arid and tropical regions where water availability is often a limiting factor, is key to sustainable production. Inoculation with AMF had increased plant establishment and survival at the seedling or transplanting stage, improved flowering and fruiting, and optimised fertiliser use, particularly phosphorus, zinc, and copper, which are immobile12,13 and critical for fruit set and quality. Several plant species of importance in horticulture and forestry are aided by AM inoculation in nursery production and then planted in the field. Abdullahi et al.14, studied the occurrence of arbuscular mycorrhizal fungi in the rhizospheres of some commonly grown medicinal plants. However, no work was conducted on AMF associated with fruit trees from the study area. Understanding AMF associated with different tree species is essential for identifying potential AMF species for inoculum production, improving orchard management, and promoting sustainable agriculture. Without such baseline data, developing microbial-based biofertilizer strategies for fruit production remains difficult. This study aims to explore the occurrence and species richness of native AMF in the rhizosphere of different fruit tree species in the University of Maiduguri Teaching and Research Farm.
MATERIALS AND METHODS
Sampling site and collection of soil samples: The study was conducted in the University of Maiduguri, located along Bama Road, Maiduguri, Borno State, Nigeria. Maiduguri is located on Latitude 11.50°N and a Longitude 13.05°E altitude of 354 m above sea level. It is usually cold and dry during Harmattan in November to January, being the coldest months, and March and April as the hottest period of the year. The mean annual temperature of the area is 32°C and the annual rainfall is between 500 and 600 mm. Sampling was done in August (rainy season), 2021. The soils and fine roots were collected from the rhizosphere of five fruit tree species: Citrus sinensis (Orange), Anacardium occidentale (Cashew), Carica papaya (Pawpaw), Musa spp. (Banana), and Mangifera indica (Mango) from the University of Maiduguri Teaching and Research Farm. A total of fifteen soil samples (3 from each plant species) were collected from 0 to 20 cm depth by pooling three sub-samples from each randomly selected tree into one composite sample using an auger. Three subsamples for each of the three replicates were collected from
the five different tree types. The samples were filled into sterile sampling bags, labelled and transported to the laboratory for further analysis. Collected soil samples were divided into two portions: One for the determination of soil physico-chemical properties and the other for AMF investigation (spore extraction and root colonization).
Soil chemical analysis: Soil samples were spread on the laboratory bench to air-dry at room temperature for 78 hrs. The air-dried soil samples were passed through 2 mm sieve and analysed for pH using pH meter in 1:2.5 (W/V) soil water suspensions. The soil particle size was determined using the hydrometer method15. Organic carbon (OC) was determined using the Walkley and Black16 wet oxidation method, and available phosphorus (AP) was determined using Bray and Kurtz17 method. Total nitrogen (%) was determined using the Kjeldahl method.
Extraction of mycorrhizal spores: The AM fungal spores were isolated from rhizosphere soil samples using the wet sieving and decanting method18, Sieves of sizes 500, 250, 150, 100, 50, and 38 μm were used for the wet sieving procedure. The sieves were arranged vertically in decreasing order of mesh sizes, with 38 μm sieve being the lowest. A 10 g of soil was dispersed in a substantial amount of water in a beaker and decanted over the sieves, followed by sucrose gradient centrifugation19. After centrifugation, spores were transferred into petri dishes and counted in three replications under a microscope (Olympus CH20i with magnification of 10×40). The spores were identified according to the descriptions and identification guidelines20. Isolated spores were identified and classified to the genus level.
AMF community studies and data analysis: Spore density (SD), species richness (SR), the frequency of occurrence (FO), and root colonization were used to study the AMF community.
| • | SD | = | Number of AMF spores/10 g soil |
| • | SR | = | Species number/soil sample |
| • | FO | = | Occurrence frequency of a specified AMF genus was defined as the percentage of the number of samples in which the genus was observed to the total number of samples |
Pearson correlation coefficient to determine the relationships between AMF spores, soil physico-chemical properties, and root colonization was adopted. All data collected were subjected to Analysis of Variance (ANOVA). Differences between treatments were separated using Fisher’s least significant difference (LSD) at 5%.
RESULTS
Physico-chemical properties of the rhizosphere soil of the different tree species: Rhizosphere soil characteristics of the different trees in the orchard as presented in Table 1 indicated that the soil was sandy loam. The highest bulk density was recorded under banana (1.52 g/cm3), and the lowest in mango (1.41 g/cm3). Rhizosphere soil of banana significantly recorded the lowest pH and OC% compared to all the tree species. However, total nitrogen and available P were significantly (p<0.05) not different in all the tree species.
| Table 1: | Physico-chemical properties of the rhizosphere’s soil from different fruit trees | |||
| Tree species | |||||
| Parameter | Banana | Mango | Cashew | Orange | Pawpaw |
| Soil type | SL | SL | SL | SL | SL |
| BD (g/cm3) | 1.52b | 1.41a | 1.42a | 1.43a | 1.51b |
| pH (1:2.5 soil- H2O) | 6.31b | 6.62a | 6.66a | 6.63 a | 6.64a |
| OC (%) | 2.16b | 2.96a | 2.85a | 2.91a | 2.80a |
| TN (%) | 0.23a | 0.23a | 0.22a | 0.20a | 0.19a |
| Avail. P (mg/kg) | 11.38a | 14.43a | 14.91a | 11.29a | 11.51a |
| Values within a row followed by the same superscript are not significantly different at p>0.05, OC: Organic carbon, TN: Total nitrogen, P: Phosphorus, SL: Sandy loam, LS: Loamy sand and L: Loam | |||||
|
|
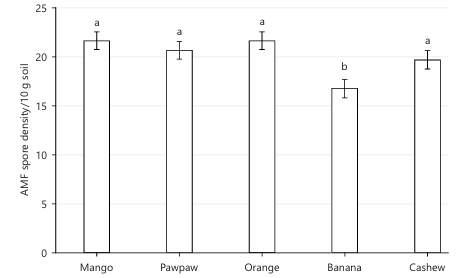
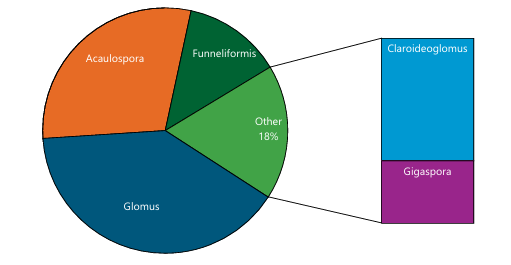
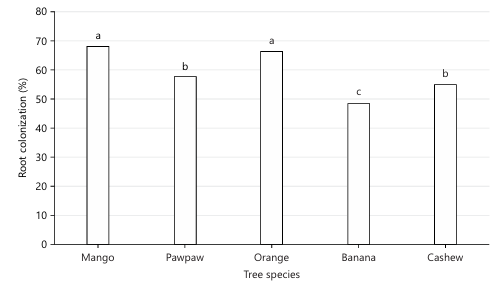
AMF community and root colonization: Figure 1-3 and Table 2 collectively demonstrate clear differences in AMF abundance, distribution, and root association among the studied fruit tree species. AMF spore density varied markedly, with banana recording the lowest density (≈12 spores/10 g soil), while mango (≈28 spores/10 g), pawpaw (≈26 spores/10 g), cashew (≈25 spores/10 g), and orange (≈24 spores/10 g) supported higher spore populations. The frequency of occurrence data revealed a strong dominance of Glomus (40%), followed by Acaulospora (29%), Funneliformis (13%), and Claroideoglomus (12%), whereas Gigaspora was least represented (6%). Root colonization also differed among tree species, with mango showing the highest colonization (69.01%), followed by orange (≈63%), pawpaw (≈61%), cashew (≈58%), and banana exhibiting the lowest colonization (49.13%). Species richness further varied across hosts, with pawpaw harboring the greatest diversity of AMF genera, while banana showed comparatively lower richness, and Gigaspora was absent in pawpaw, indicating host-specific AMF associations.
Relationship between soil properties and AMF community: The Pearson correlation coefficient, as presented in Table 3, indicated organic carbon showed a strong positive correlation with both spore density (r = 0.52) and % root colonization (r = 0.73). Available P and bulk density were negatively correlated, indicating that higher P and bulk density could reduce AM fungal population. A weak positive correlation was observed between pH and the fungal community. Spore density and % root colonization was strongly correlated since % colonization increases with increase in spore density.
|
| Table 2: | Species richness (SR) of abuscular mycorrhizal fungi under different tree species | |||
| Tree species | Acaulospora | Glomus | Gigasspora | Claroideoglomus | Funneliformis |
| Mango | 5 | 10 | 2 | 3 | 2 |
| Pawpaw | 7 | 8 | - | 2 | 4 |
| Orange | 7 | 8 | 2 | 2 | 3 |
| Banana | 6 | 6 | 1 | 2 | 2 |
| Cashew | 5 | 9 | 1 | 3 | 2 |
| Table 3: | Pearson’s correlation coefficient between soil properties and AMF community | |||
| Parameter | BD (g/cm3) | pH (H2O) | OC (%) | TN (%) | Avail.P (mg/kg) | Spore density | Colonization (%) |
| BD (g/cm3) | 1 | -0.47 | -0.52 | -0.31 | 0.29 | -0.45 | -0.68* |
| pH (H O) | 1 | 0.46 | 0.28 | -0.22 | 0.39 | 0.42 | |
| OC (%) | 1 | 0.36 | -0.4 | 0.52 | +0.73* | ||
| TN (%) | 1 | 0.11 | 0.1 | 0.08 | |||
| Avail. P (mg/kg) | 1 | -0.41 | -0.61* | ||||
| Spore density | 1 | +0.66* | |||||
| Colonization (%) | 1 | ||||||
| *Significant correlations at p<0.05, BD: Bulk density (g/cm3), pH: Soil reaction measured in water (1:2.5), OC: Organic carbon (%), TN: Total nitrogen (%), Avail. P: Available phosphorus (mg/kg), values represent Pearson’s correlation coefficients (r), indicating the strength and direction of relationships between soil properties and AMF parameters, Positive values indicate positive correlations, while negative values indicate inverse relationships and Asterisks (*) denote statistically significant correlations at p<0.05 | |||||||
DISCUSSION
The present study has confirmed the presence of arbuscular mycorrhizal fungi associated with the five fruit tree species planted in the University of Maiduguri Orchard. Several researchers have similarly reported the symbiotic relationship of AMF with orange tree21-23, mango24, banana25, pawpaw26 and cashew27. Positive impact of AMF inoculants on growth and survival of several fruit tree species during nursery and field phases was documented28,29. AMF colonization varied among the tree species. Mango recorded the highest % root colonization (69.01%) while banana recorded the least (49.13%). These discrepancies in the colonization of AMF may be attributed to mycorrhizal dependencies of the different tree species30 or the colonization potential of various AMF species31,32. In addition, trees contribute organic matter to the soil in different ways, through mucilage, dead/decaying root tissues, fallen leaves, and root exudates released into the rhizosphere33. These inputs provide energy for many soil organisms that drive biological activity and interactions, ultimately influencing soil nutrient levels and overall fertility.
In our study, Glomus and Acaulospora were the predominant genera among AMF communities observed. A similar trend was also noted by Abdullahi et al.14 they revealed Glomus genera as the predominant species associated with some medicinal plants in the same study area, followed by
Acaulospora. Wang et al.33 investigated AMF associated with tree species in a planted forest of eastern China and reported Glomus and Acaulospora as the dominant genera. This trend was extensively reported in both natural and agricultural ecosystems across the globe34-36. Dominant occurrence of Glomus genera over other AMF species in several fruit trees has been documented26,37,38. The dominance of Glomus species in the study area indicates that this genus is highly adapted to the prevailing environmental conditions and can thrive across various ecological niches. Fabre et al.37 explained that this dominance may result from these fungi producing far more spores in a shorter time than other genera. Alternatively, their prevalence might reflect the genus’s strong ability to cope with harsh conditions such as drought, salinity, and other environmental stresses or its capacity to thrive across many different ecological environments38 Furthermore, arbuscular mycorrhizal fungi from the genera Glomus and Acaulospora are known for their resilience to soil disturbances and environmental changes39. Therefore, understanding which AMF species occur in the rhizosphere and roots of naturally growing plants is important for identifying the most common and environmentally resilient species suitable for inoculum formulation.
This study observed that available phosphorus and bulk density had clear negative correlations with AMF colonization and spore density, indicating that excessive P or compaction reduces AM fungal proliferation. It is well established that soil characteristics significantly influence AMF colonization. Elevated soil fertility can negatively impact the ability of AMF to sporulate and colonize roots40. Among the essential nutrients, phosphorus (P) has the strongest link with mycorrhizal development, as it plays a key role in controlling AMF colonization rates30. Our findings corroborated those of previous studies41,42. They observed a decrease in AMF colonization in soils with high phosphate concentrations43. Sakha et al.44 have shown that AMF richness decreased with increasing soil available P and increased with soil pH. In this study, variations in the physical and chemical properties of rhizosphere soil may have resulted from differences among tree species, which in turn could influence AMF colonization and community structure.
The results from our study also revealed that soil organic carbon and pH are key positive drivers of AMF activity. Soil pH supported higher fungal activity, showing a mild positive relationship. Organic carbon showed the strongest positive correlation with both spore density and % colonization. This aligned with previous studies34. They reported soil pH and soil organic carbon as the main factors influencing the AMF community. Muzakir45 observed increased AMF spore numbers and species diversity as the organic matter and soil pH increased. He thus inferred that the amount and type of mycorrhizal spores were affected by the soil chemistry. On the other hand, variation from the allocation of C from the host plant and exudation from roots to the soil could be beneficial for the sporulation of AM fungi46.
CONCLUSION
The present study confirmed the association of Arbuscular Mycorrhizal Fungi (AMF) with five fruit tree species in the University of Maiduguri Orchard, demonstrating variations in colonization intensity among species. Mango exhibited the highest root colonization, while banana recorded the lowest, reflecting differences in host mycorrhizal dependency and AMF colonization potential. The predominance of the genera Glomus and Acaulospora aligns with global trends and indicates their ecological adaptability to the prevailing environmental conditions of the study area. Soil properties were found to play a significant role in shaping AMF activity, with available phosphorus and bulk density exerting negative effects, whereas soil organic carbon and pH showed strong positive correlations with spore density and root colonization. These findings highlight the importance of maintaining optimal soil conditions for AMF development and underscore the potential of dominant, resilient species such as Glomus and Acaulospora for inoculum formulation. Harnessing these fungi could enhance fruit tree growth, nutrient uptake, and soil health, thereby contributing to sustainable orchard management and improved fruit yield.
SIGNIFICANCE STATEMENT
This study discovered the dominant and ecologically resilient arbuscular mycorrhizal fungi associated with selected fruit tree species, particularly Glomus and Acaulospora, which can be beneficial for sustainable orchard management and biofertilizer development. By elucidating the relationships between soil properties and AMF colonization, the findings provide a scientific basis for reducing dependence on chemical fertilizers while improving soil fertility and crop productivity. This study will help researchers to uncover critical areas of host soil microbe interactions that many researchers were not able to explore. Thus, a new theory on AMF-driven, soil-based sustainable fruit production may be arrived at.
REFERENCES
- Fernández, N.V., M.I. Messuti and S.B. Fontenla, 2013. Occurrence of arbuscular mycorrhizas and dark septate endophytes in pteridophytes from a Patagonian Rainforest, Argentina. J. Basic Microbiol., 53: 498-508.
- Smith, S.E. and D.J. Read, 2008. Mycorrhizal Symbiosis. 3rd Edn., Academic Press, New York, USA, ISBN: 978-0-12-370526-6, Pages: 787.
- Davison, J., M. Moora, M. Öpik, A. Adholeya and L. Ainsaar et al., 2015. Global assessment of arbuscular mycorrhizal fungus diversity reveals very low endemism. Science, 349: 970-973.
- Hartoyo, B. and O. Trisilawati, 2021. Diversity of arbuscular mycorrhiza fungi (AMF) in the rhizosphere of sugarcane. IOP Conf. Ser.: Earth Environ. Sci., 653.
- Zhang, B., F. Shi, X. Zheng, H. Pan, Y. Wen and F. Song, 2023. Effects of AMF compound inoculants on growth, ion homeostasis, and salt tolerance-related gene expression in Oryza sativa L. under salt treatments. Rice, 16.
- Abdel-Salam, E., A. Alatar and M.A. El-Sheikh, 2018. Inoculation with arbuscular mycorrhizal fungi alleviates harmful effects of drought stress on damask rose. Saudi J. Biol. Sci., 25: 1772-1780.
- Seemakram, W., T. Suebrasri, S. Khaekhum, J. Ekprasert, T. Aimi and S. Boonlue, 2021. Growth enhancement of the highly prized tropical trees siamese rosewood and burma padauk. Rhizosphere, 19.
- Ma, J., Q. Zhao, S. Zaman, A. Anwar and S. Li, 2024. The transcriptomic analysis revealed the molecular mechanism of arbuscular mycorrhizal fungi (AMF) inoculation in watermelon. Sci. Hortic., 332. .
- Hussain, M., M. Adeel and J.C. White, 2025. Nano-selenium: A novel candidate for plant microbiome engineering. Trends Plant Sci., 30: 454-456.
- Jia, T., Y. Zhang, Y. Yao, Y. Wang and X. Liang et al., 2023. Effects of AMF inoculation on the eco-physiological characteristics of Imperata cylindrica under differing soil nitrogen conditions. Front. Plant Sci., 14.
- Liu, W., Y. Zhang, S. Jiang, Y. Deng and P. Christie et al., 2016. Arbuscular mycorrhizal fungi in soil and roots respond differently to phosphorus inputs in an intensively managed calcareous agricultural soil. Sci. Rep., 6.
- Haougui, A. P.S. Souniabe, A. Doumma and T. Adam, 2013. Evolution of endomycorrhizal fungal populations on weeds in four market garden sites in the Maradi Region of Niger [In French]. Int. J. Biol. Chem. Sci., 7: 554-565.
- Bossou, L.D.R., H.B. Houngnandan, A. Adandonon, C. Zoundji and P. Houngnandan, 2019. Diversity of arbuscular mycorrhizal fungi associated with maize (Zea mays L.) cultivation in Benin [In French]. Int. J. Biol. Chem. Sci., 13: 597-609.
- Abdullahi, R., J.S. Kwari and A.M. Zubairu, 2023. Determination of arbuscular mycorrhizal fungi (AMF) association with some selected medicinal plants. Res. Highlights Agric. Sci., 9: 71-80.
- Bouyoucos, G.J., 1962. Hydrometer method improved for making particle size analyses of soils. Agron. J., 54: 464-465.
- Walkley, A. and I.A. Black, 1934. An examination of the degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci., 37: 29-38.
- Bray, R.H. and L.T. Kurtz, 1945. Determination of total, organic and available forms of phosphorus in soils. Soil Sci., 59: 39-46.
- Gerdemann, J.W. and T.H. Nicolson, 1963. Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting. Trans. Br. Mycol. Soc., 46: 235-244.
- Ianson, D.C. and M.F. Allen, 1986. The effects of soil texture on extraction of vesicular-arbuscular mycorrhizal fungal spores from arid sites. Mycologia, 78: 164-168.
- Huang, H., X. Qin, Y. Kang, J. Xu and P. Shang et al., 2025. Three new species and a new record of arbuscular mycorrhizal fungi of the genus Acaulospora associated with citrus from South China. J. Fungi, 11.
- Wu, Q.S., A.K. Srivastava, Y.N. Zou and S.K. Malhotra, 2017. Mycorrhizas in citrus: Beyond soil fertility and plant nutrition. Indian J. Agric. Sci., 87: 427-443.
- Ishaq, L., A.V. Simamora, P.O. Bako, Y.I. Benggu, M.M. Airthur, E. Roefaida and E.S.O. Nguru, 2023. Abundance of arbuscular mycorrhizal fungi in the rhizosphere of healthy and declining citrus in East Nusa Tenggara, Indonesia. Int. J. Biol. Chem. Sci., 7.
- Yang, C., Z. Teng, Z. Jin, Q. Ouyang and L. Lv et al., 2025. Structure and composition of arbuscular mycorrhizal fungal community associated with mango. Front. Plant Sci., 16.
- Mahecha-Vásquez, G., S. Sierra and R. Posada, 2017. Diversity indices using arbuscular mycorrhizal fungi to evaluate the soil state in banana crops in Colombia. Appl. Soil Ecol., 109: 32-39.
- Muiruri, J., F.K. Rimberia, M.R. Mwashasha and A. Kavoo, 2022. Abundance and diversity of arbuscular mycorrhizal gungal (AMF) spores isolated from the rhizosphere of papaya and other different cropping systems in Central Kenya. J. Agric. Sci. Technol., 21: 18-36.
- Suada, I.K., E. Prima, M. Sritamin, I.W. Adiartayasa, I.G.K. Susrama and I.G.P. Wirawan, 2018. Isolation and identification of arbuscular mycorrhizal fungi (AMF) in cashew plants (Anacardium occindentale L.) in Datah Village, Abang District of Karangasem Regency. Int. J. Biosci. Biotechnol., 5: 168-175.
- Mapunda, H.B., S. Declerck, K.M. Mtei and A.I. Meya, 2025. Arbuscular mycorrhizal fungi inoculation enhanced banana plant growth and fruit yield in Central-Northern Tanzania. Discover Sustainability, 6.
- Ortas, İ., M. Rafique, C. Akpinar and Y.A. Kacar, 2017. Growth media and mycorrhizal species effect on acclimatization and nutrient uptake of banana plantlets. Sci. Hortic., 217: 55-60.
- Kowalska, I., A. Konieczny, M. Gąstoł, W. Sady and E. Hanus-Fajerska, 2015. Effect of mycorrhiza and phosphorus content in nutrient solution on the yield and nutritional status of tomato plants grown on rockwool or coconut coir. Agric. Food Sci., 24: 39-51.
- Minggui, G., T. Ming, Z. Qiaoming and F. Xinxin, 2012. Effects of climatic and edaphic factors on arbuscular mycorrhizal fungi in the rhizosphere of Hippophae rhamnoides in the Loess Plateau, China. Acta Ecol. Sin., 32: 62-67.
- Kim, S.J., J.K. Eo, E.H. Lee, H. Park and A.H. Eom, 2017. Effects of arbuscular mycorrhizal fungi and soil conditions on crop plant growth. Mycobiology, 45: 20-24.
- Bertin, C., X. Yang and L.A. Weston, 2003. The role of root exudates and allelochemicals in the rhizosphere. Plant Soil, 256: 67-83.
- Wang, J., G.G. Wang, B. Zhang, Z. Yuan and Z. Fu et al., 2019. Arbuscular mycorrhizal fungi associated with tree species in a planted forest of Eastern China. Forests, 10.
- Trejo, D., I. Barois and W. Sangabriel-Conde, 2016. Disturbance and land use effect on functional diversity of the arbuscular mycorrhizal fungi. Agrofor. Syst., 90: 265-279.
- Xiang, D., E. Verbruggen, Y. Hu, S.D. Veresoglou and M.C. Rillig et al., 2014. Land use influences arbuscular mycorrhizal fungal communities in the farming-pastoral ecotone of Northern China. New Phytol., 204: 968-978.
- Silvana, V.M., F.J. Carlos, A.C. Lucía, A. Natalia and C. Marta, 2018. Colonization dynamics of arbuscular mycorrhizal fungi (AMF) in Ilex paraguariensis crops: Seasonality and influence of management practices. J. King Saud Univ. Sci., 32: 183-188.
- Fabre, F., J.B. Burie, A. Ducrot, S. Lion, Q. Richard and R. Djidjou-Demasse, 2022. An epi-evolutionary model for predicting the adaptation of spore-producing pathogens to quantitative resistance in heterogeneous environments. E Appl., 15: 95-110.
- Houngnandan, P., F. Zapata, P. Boekx and O. van Cleemput, 2010. Effect of Bradyrhizobial inoculation and N fertilization on promiscuous soybean and subsequent maize yield grown in degraded “terre de barre” in Benin. Ann. Sci. Agronomiques, 12.
- Welemariam, M., F. Kebede, B. Bedadi and E. Birhane, 2018. Effect of community-based soil and water conservation practices on arbuscular mycorrhizal fungi types, spore densities, root colonization, and soil nutrients in the northern highlands of Ethiopia. Chem. Biol. Technol. Agric., 5.
- Liu, X., Z. Feng, H. Zhu and Q. Yao, 2019. Exogenous abscisic acid and root volatiles increase sporulation of Rhizophagus irregularis DAOM 197198 in asymbiotic and pre-symbiotic status. Mycorrhiza, 29: 581-589.
- Zeng, H., L. Yu, P. Liu, Z. Wang, Y. Chen and J. Wang, 2021. Nitrogen fertilization has a stronger influence than cropping pattern on AMF community in maize/soybean strip intercropping systems. Appl. Soil Ecol., 167.
- van der Heijden, M.G.A., J.N. Klironomos, M. Ursic, P. Moutoglis and R. Streitwolf-Engel et al., 1998. Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature, 396: 69-72.
- Datta, P. and M. Kulkarni, 2012. Arbuscular mycorrhizal fungal diversity in sugarcane rhizosphere in relation with soil properties. Notulae Sci. Biol., 4: 66-74.
- Sakha, M., J.P. Gweyi-Onyango, C. Masso and F.P. Baijukya, 2025. Diversity, characteristics, and abundance of native arbuscular mycorrhizal fungi in the semi-arid lands of Eastern Kenya. Front. Microbiol., 16.
- Muzakkir, 2011. The relationship between indigeneous arbuscular mycorrhazal funds and soil chemical properties in the critical land of Tanjung Alai, West Sumatra [In Indonesian]. J. Solum, 8: 53-57.
- Ba, L., J. Ning, D. Wang, E. Facelli, J.M. Facelli, Y. Yang and L. Zhang, 2012. The relationship between the diversity of arbuscular mycorrhizal fungi and grazing in a meadow steppe. Plant Soil, 352: 143-156.
How to Cite this paper?
APA-7 Style
Abdullahi,
R., F.,
M., A.,
K.H. (2026). Arbuscular Mycorrhizal Fungi in the Rhizosphere of Selected Fruit Tree Species in Maiduguri, Nigeria. Trends in Biological Sciences, 2(1), 99-107. https://doi.org/10.21124/tbs.2026.99.107
ACS Style
Abdullahi,
R.; F.,
M.; A.,
K.H. Arbuscular Mycorrhizal Fungi in the Rhizosphere of Selected Fruit Tree Species in Maiduguri, Nigeria. Trends Biol. Sci 2026, 2, 99-107. https://doi.org/10.21124/tbs.2026.99.107
AMA Style
Abdullahi
R, F.
M, A.
KH. Arbuscular Mycorrhizal Fungi in the Rhizosphere of Selected Fruit Tree Species in Maiduguri, Nigeria. Trends in Biological Sciences. 2026; 2(1): 99-107. https://doi.org/10.21124/tbs.2026.99.107
Chicago/Turabian Style
Abdullahi, Rakiya, Maina F., and Kellu H. A..
2026. "Arbuscular Mycorrhizal Fungi in the Rhizosphere of Selected Fruit Tree Species in Maiduguri, Nigeria" Trends in Biological Sciences 2, no. 1: 99-107. https://doi.org/10.21124/tbs.2026.99.107

This work is licensed under a Creative Commons Attribution 4.0 International License.




