Phytochemical Profiling and GC-MS Analysis of Conocarpus lancifolius Fruits
| Received 30 Oct, 2025 |
Accepted 24 Jan, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Plants are repositories of several natural chemicals; the identification of which are important in the field of medicine, cosmetics, health and other industries. Conocarpus lancifoliusEngl.(Family-Combretaceae) is an evergreen tree cultivated as an ornamental plant species. Since no comprehensive phytochemical analysis of its fruits was available, an attempt was made to evaluate the phytochemical composition and phyto-constituents present in its methanolic fruit extract. Materials and Methods: Both dry (mature) and green (immature) fruits (Dfr and Gfr, respectively) were collected from Udaipur city, Rajasthan, India. The aqueous and methanolic extracts underwent preliminary qualitative phytochemical screening, while the methanolic extract was subjected to Gas Chromatography-Mass Spectrometry (GC-MS) analysis for the first time. Results: The preliminary screening revealed the presence of carbohydrates, flavonoids, phenols, cardiac glycosides, terpenoids, coumarins, tannins, steroids, and phlobatannins, and the absence of amino acids and saponins. The GC-MS analysis demonstrated the presence of 80 and 65 biological compounds in Dfr and Gfr, respectively. The major compounds identified in Dfr and Gfr were, 1,2,3-benzenetriol (13.19, 26.13%), n-hexadecanoic acid (15.92, 11.12%), β-sitosterol (10.84, 9.57%), 10(E),12(Z)-Conjugated linoleic acid (9.51, 2.66%) and Lupeol (5.09, 5.57%), respectively. Conclusion: The preliminary phytochemical analysis and GC-MS profiling of C. lancifolius fruits from India have shown the presence of biologically significant phytochemicals for the first time and could stimulate future research to evaluate its complete pharmacological potential.
| Copyright © 2026 Mittal and Jain. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Metabolites are small molecules produced during metabolic processes and can be classified as primary or secondary. Primary metabolites, such as vitamins, amino acids, nucleotides, and organic acids, are essential for growth, reproduction, and normal physiological functions. In contrast, secondary metabolites, synthesized mainly during the stationary growth phase, are not directly involved in growth but play vital roles in plant defense and adaptation found abundantly in medicinal plants1. Moreover, the secondary metabolites, for example, alkaloids, flavonoids, tannins, phenolics, and saponins, exhibit diverse biological activities with significant therapeutic value2.

|
A qualitative and quantitative analysis of the phyto-constituents offers valuable insights for the discovery of new drugs. Gas Chromatography-Mass Spectrometry (GC-MS) is a reliable analytical method that has been widely used for the structural elucidation of a broad range of phyto-constituents, such as glycosides, flavonoids, phenolics, essential oils, alkaloids, saponins, steroids, and their derivatives, in addition to the analysis of volatile compounds3. Thus, the GC-MS profiling of a plant extract helps in the identification of pharmacologically valuable compounds.
Conocarpus lancifolius Engl. (Fig. 1) belonging to the family Combretaceae is an ornamental evergreen tree native to Somalia. It is commonly known as Lanceleaf Buttonwood and Damas tree and is notable for its resistance to heat and salt, as well as its tolerance to drought4. The plant is under the vulnerable category as per the IUCN Red Data List, and its decreasing population trend is a matter of concern5. Aerial parts, leaves, roots, and fruits of this plant have been explored for their phyto-pharmaceutical potential, and several pharmacological activities, including antioxidant, antimicrobial, anxiolytic, antidiabetic, cytotoxic, antiurease, cardio-protective, antiquorum-sensing, and acetylcholinesterase inhibition have been reported6-14. Besides, many secondary metabolites like phenolic compounds, flavonoids, alkaloids, fatty acids, steroids, terpenoids, coumarins, saponins, tannins, glycosides, anthraquinones etc. have been identified from leaves, roots, stems, and aerial parts of C. lancifolius.
Al-Taweel et al.6 have isolated one novel compound 3,3’4' trimethoxy 4-O-cyclopentanone ellagic acid, and two known compounds, kaempferol 3-O-rutinose and β sitosterol 3-O-glucoside, from the fruits of C. lancifolius. Whereas Afifi et al.7 have reported several polyphenolic compounds from ethanolic fruit extract of C. lancifolius, such as 4-hydroxy benzoic acid, vanillic acid, caffeic acid, 1,2-dihydroxy benzene, catechin, benzoic acid, p-coumaric acid, t-ferulic acid, sinapic acid, vanillin, rutin hydrate, cinnamic acid, protocatechuic acid, quercetin, and tannins. However, phytochemical investigationofto its fruits through GC-MS have still not been carried out. Hence, the present study was undertaken to investigate the bioactive compounds present in the methanolic extract of Conocarpus lancifolius fruits, contributing to a better understanding of its phytochemical composition.
MATERIALS AND METHODS
Plant collection and identification: The dry (fully mature) and green (immature) fruits of C. lancifolius were collected during February-March, 2025 from Udaipur City, Rajasthan, India. The voucher specimen was prepared and authentication of the plant was carried out at Arid Zone Regional Centre, Botanical Survey of India (BSI), Jodhpur (No./BSI/AZRC/A.12012/Tech./2025-26(Pl. Id.)/445; dated 17.09.2025).
Preparation of plant extracts: Both dry and green fruits were washed under running tap water and air-dried in the shade at room temperature. After drying, both were powdered separately, coded as Dfr and Gfr, respectively, and stored at 4°C in a refrigerator. For preliminary qualitative phytochemical and GC-MS analyses, the following two types of extracts were prepared from the powdered plant materials.
Aqueous extract: Aqueous extracts were freshly prepared by soaking 400 mg of each Dfr and Gfr of C. lancifolius in 20 mL of distilled water separately, followed by boiling for 20 min. These were cooled and filtered through Whatman’s No. 1 filter paper. The clear filtrates were utilized instantly for preliminary qualitative phytochemical screening.
Methanolic extract: One hundred gram of both Dfr and Gfr were soaked in 700 mL of methanol separately for eight days with periodic stirring and then filtered using Whatman’s filter paper No. 1. The filtrates were evaporated in a boiling water bath at 40°C, and the concentrated extracts were stored in sterile glass petri dishes at 4ºC in the refrigerator. The percent yields obtained for methanolic extracts of Dfr and Gfr were 1.88 and 10.34%, respectively. These extracts were used for qualitative phytochemical assessment as well as analyzed for the presence of various phytochemicals through GC-MS.
Qualitative phytochemical screening: Aqueous/ME or dry and green fruit powders of C. lancifolius were used for preliminary qualitative phytochemical screening of amino acids, carbohydrates, terpenoids, steroids, cardiac glycosides, phlobatannins, flavonoids, phenols, tannins, coumarins, and saponins as required. These tests were performed as per standard methodology15.
Gas Chromatography-Mass Spectrometry (GC-MS) analysis: To find out the chemical compounds present in the methanolic extracts of Dfr and Gfr of C. lancifolius, Gas Chromatography-Mass Spectrometry analysis was conducted on a Shimadzu GC-MS-QP2010 Ultra system with an AOC-20i+s autosampler. The system employed a Rxi-5 SIL MS column (30 m×0.25 mm i.d×0.25 mm film thickness) with helium as the carrier gas at a constant flow rate of 1.21 mL/min. In split mode, the injector operated with a 10:1 split ratio, 1 μL of injection volume, and 270°C injection temperature. The column oven temperature was initially established at 70°C for 2 min, subsequently increased at a rate of 10°C/min to 300°C, and held for 15 min. Ion source and interface temperatures were maintained at 220°C and 280°C, respectively. In scan mode, mass spectra were obtained with a solvent cut-off time of 3.50 min, a mass range of 40-600 m/z, and a scan rate of 3333 scans per second. Data were acquired in Total Ion Count (TIC) mode. Identifications of compounds were made by matching retention times and mass spectral patterns against the National Institute of Standards and Technology (NIST) and Wiley libraries. The relative abundance of each analyte was computed as the percentage of its peak area relative to the aggregate peak area of all detected components.
RESULTS
Qualitative phytochemical analysis: A preliminary qualitative phytochemical analysis of both dry and green fruits of C. lancifolius has shown the presence of one primary metabolite that is carbohydrate and eight secondary metabolites, namely, flavonoid, phenol, cardiac glycoside, terpenoid, coumarin, steroid, tannin and phlobatanin. One primary metabolite amino acid and one secondary metabolite saponin were not detected in both Dfr and Gfr (Table 1).
GC-MS analysis: The methanolic extracts of dry and green fruits of C. lancifolius, analyzed through GC-MS, revealed 84 and 71 peaks in the chromatograms, corresponding to 80 and 65 compounds, respectively (Fig. 2 and 3). Among which, n-hexadecanoic acid (15.92%), pyrogallol (13.19%), β-sitosterol (10.84%), 10(E),12(Z)-Conjugated linoleic acid (9.51%), Oleic acid (5.36%) and Lupeol (5.09%) in Dfr (Table 2) whereas pyrogallol (26.13%), n-hexadecanoic acid (11.12%), β-sitosterol (9.57%) and Lupeol (5.57%) in Gfr (Table 3), were the most abundant compounds. Among the variety of secondary metabolites in C. lancifolius, terpenoids and fatty acid derivatives are predominant compounds present, followed by phenolic compounds, phytosterols, hydrocarbons, heterocyclic compounds, and other miscellaneous compounds (Table 4).
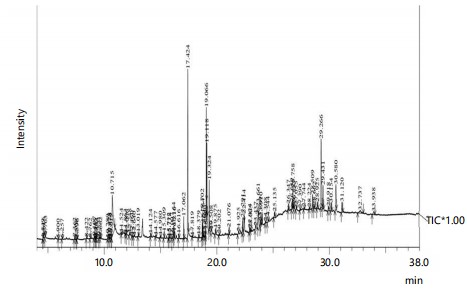
|
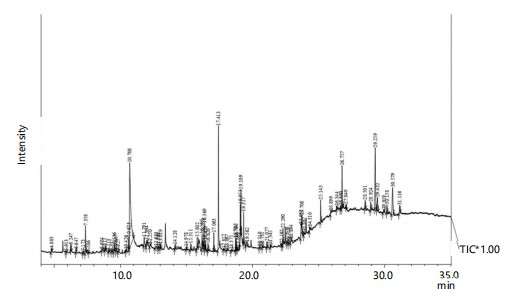
|
The compounds which are unique to dry mature fruits of C. lancifolius are Diglycolic acid, ethyl 2-isopropoxyphenyl ester, Octanoic acid, Phenol, 8-Hydroxy-2-octanone, 2,4,6-Cycloheptatrien-1-one, Dehydromevalonic lactone, Dianhydromannitol, 3-Dimethylsilyloxytetradecane, Nonanoic acid, Acetic acid, 1,3,7-trimethylocta-2,6-dienyl ester, Dodecanal dimethyl acetal, 3-Methyl-2-(2-methyl-2-butenyl)- furan, 2-Isopropenyl-3-methylpyrazine, Guaia-6,9-diene, Longifolene, 6-Dimethyl(trimethylsilyl)silyloxytetradecane, D-Mannoheptulose, γ-Elemene, α-Amorphene, Dodecanoic acid, Canophyllal, Orcinol, monoacetate, Decane, 2,3,8-trimethyl-, Isopropyl palmitate, 2-Methyl tetracosane, Heptadecanoic acid, Oleic Acid, Undec-10-ynoic acid, tetradecyl ester, Eicosanoic acid, 1H-1,3-Benzimidazol-7-amine, N-[(4-methoxyphenyl)methyl, Tyrosine, 9-Hexacosene, 1-Decyloxymethyl-3-methyl-1,3-dihydrobenzoimidazol-2-ylideneamine, Ethyl 6,9,12,15-octadecatetraenoate, 9,12-Octadecadienoic acid (Z,Z)-, 2,3-dihydroxypropyl ester, Octadecanoic acid, 2,3-dihydroxypropyl ester, Benzoic acid, 2,4-dihydroxy-, 4-(1,1-dimethylpropyl)phenyl, Oleamide, 1,54-dibromotetrapentacontane, Dodecane, 1,1-dimethoxy-, Tetracosan-10-yl acetate, Campesterol, Stigmast-5-en-3-ol, oleat, Stigmasta-4,22-diene, Stigmastane-3,6-dione, (5-alpha), α-amyrin and Cyclopentanone, 2-(5-oxohexyl) as shown in Table 2. Besides, two peaks were obtained for four compounds, namely, 9-Octadecenamide, Phenol, 2-methyl-5-(1-methylethyl), Neophytadiene, and 10(E),12(Z)-Conjugated linoleic acid (Table 2).
| Table 1: | Qualitative phytochemical analysis of fruits of Conocarpus lancifolius Engl. | |||
| Phytochemical | Dry fruit (Dfr) | Green fruit (Gfr) |
| Carbohydrate | + | + |
| Amino acid | - | - |
| Flavonoid | + | + |
| Phenol | + | + |
| Cardiac glycoside | + | + |
| Terpenoid | + | + |
| Coumarin | + | + |
| Steroid | + | + |
| Saponin | - | - |
| Tannin | + | + |
| Phlobatanin | + | + |
| +: Present and -: Absent | ||
| Table 2: | Phyto-compounds identified through GC-MS in methanolic extract of Conocarpus lancifolius dry fruits (Dfr) | |||
| Peak | R. Time | Area % | Name of the compound | Molecular formula |
Molecular weight (g/mol) |
Name of compound |
| 1 | 4.549 | 0.05 | Diglycolic acid, ethyl 2-isopropoxyphenyl ester |
C15H20O6 | 296 | Aromatic ester |
| 2 | 4.616 | 0.06 | Octanoic acid (Caprylic acid) | C8H16O2 | 144 | Saturated fatty acid |
| 3 | 4.763 | 0.15 | Phenol | C6H5OH | 94 | Phenolic compound |
| 4 | 5.9 | 0.01 | 4-hydroxy-2,5-dimethyl- 3(2h)-furanone |
C6H8O3 | 128 | Heterocyclic compound |
| 5 | 6.257 | 0.07 | 8-Hydroxy-2-octanone | C8H16O2 | 144 | Aliphatic hydroxyl ketone |
| 6 | 7.362 | 0.23 | 4H-Pyran-4-one, 2,3-dihydro-3, 5-dihydroxy-6-methyl (Pyranone) |
C6H8O4 | 144 | Phenolic compound |
| 7 | 7.508 | 0.02 | 2,4,6-Cycloheptatrien-1- one (Tropone) |
C7H6O | 106 | Conjugated ketone |
| 8 | 7.596 | 0.13 | Dehydromevalonic lactone | C6H8O2 | 112 | Cyclic α,β-unsaturated lactone |
| 9 | 8.422 | 0.16 | Dianhydromannitol | C6H10O4 | 146 | Sugar alcohol derivative |
| 10 | 8.762 | 0.06 | 3-Dimethylsilyloxytetradecane | C16H36OSi | 272 | Alkane derivative |
| 11 | 9.107 | 0.18 | Nonanoic acid | C9H18O2 | 158 | Fatty Acid |
| 12 | 9.185 | 0.03 | Acetic acid, 1,3,7-trimethylocta- 2,6-dienyl ester |
C13H22O2 | 210 | Monoterpene ester |
| 13 | 9.262 | 0.02 | Dodecanal dimethyl acetal | C14H30O2 | 230 | Fatty acid |
| 14 | 9.333 | 0.08 | Phenol, 2-methyl-5-(1-methylethyl), (O-Thymol/Carvacrol) |
C10H14O | 150 | Phenolic compound |
| 15 | 9.531 | 0.37 | 3-Methyl-2-(2-methyl-2-butenyl)- furan (Rosefuran) |
C10H14O | 150 | Terpenoid |
| 16 | 9.662 | 0.16 | Phenol, 2-methyl-5-(1-methylethyl), (O-Thymol/Carvacrol) |
C10H14O | 150 | Phenolic compound |
| 17 | 10.289 | 0.02 | 2-Isopropenyl-3-methylpyrazine | C8H10N2 | 134 | Heterocyclic compound |
| 18 | 10.423 | 0.05 | Phenol, 2-methoxy-4-(2-propenyl, (1,3,4-Eugenol) |
C10H12O2 | 64 | Phenolic compound |
| 19 | 10.478 | 0.07 | n-Decanoic acid (Capric acid ) | C10H20O2 | 172 | Fatty Acid |
| 20 | 10.619 | 0.04 | 4-tert-Butylcyclohexyl acetate | C12H22O2 | 198 | Acid ester |
| 21 | 10.715 | 13.19 | 1,2,3-benzenetriol (Pyrogallol) | C6H3(OH)3 | 126 | Phenolic compound |
| 21 | 11.524 | 0.13 | Guaia-6,9-diene | C15H24 | 204 | Sesquiterpene |
| 23 | 11.863 | 0.07 | Longifolene | C15H24 | 204 | Sesquiterpene |
| 24 | 12.006 | 0.07 | 6-Dimethyl(trimethylsilyl)silyloxytetradecane | C19H44OSi2 | 344 | Organosilane |
| 25 | 12.248 | 0.28 | D-Mannoheptulose | C7H14O7 | 210 | Monosaccharide |
| 26 | 12.46 | 0.07 | γ-Elemene | C15H24 | 204 | Sesquiterpene |
| 27 | 12.64 | 0.15 | α-Amorphene | C15H24 | 204 | Sesquiterpene |
| 28 | 13.019 | 0.38 | Dodecanoic acid (Lauric acid) | C12H24O2 | 200 | Fatty Acid |
| 29 | 14.124 | 0.37 | Isocitronellol | C10H20O | 156 | Monoterpenoid |
| 30 | 14.577 | 0.23 | Caryophyllane, 4,8- beta-epoxy | C15H26O | 222 | Sesquiterpene |
| 31 | 14.98 | 0.29 | Canophyllal | C30H48O2 | 440 | Pentacyclic triterpenoid |
| 32 | 15.309 | 0.48 | Tetradecanoic acid (Myristic acid) | C14H28O2 | 228 | Fatty Acid |
| 33 | 15.712 | 0.1 | Orcinol, monoacetate | C7H8O2 | 124 | Phenolic compounds |
| 34 | 15.778 | 0.21 | Decane, 2,3,8-trimethyl- | C13H28 | 184 | Branched alkane |
| 35 | 16.007 | 0.17 | Isopropyl palmitate | C19H38O2 | 298 | Fatty acid ester |
| 36 | 16.164 | 0.67 | Neophytadiene | C20H38 | 278 | Diterpene |
| 37 | 16.216 | 0.33 | 2L, 4d-dihydroxyeicosane | C20H42O2 | 314 | Fatty alcohol |
| 38 | 16.616 | 0.4 | Phytol (3,7,11,15-Tetramethyl- 2-hexadecen-1-ol) |
C20H42O | 296 | Diterpenoid |
| 39 | 17.062 | 1.47 | Hexadecanoic acid, methyl ester (Palmitic acid methyl ester) |
C17H34O2 | 270 | Fatty acid ester |
| 40 | 17.424 | 15.92 | n-Hexadecanoic acid (Palmitic acid) | C16H32O2 | 256 | Fatty acid ester |
| 40 | 17.819 | 0.09 | 2-Methyl tetracosane | C25H52 | 352 | Alkane |
| 42 | 18.379 | 0.25 | Heptadecanoic acid (Margaric acid) | C17H34O2 | 270 | Saturated fatty acid |
| 43 | 18.702 | 1.78 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester (Methyl linoleate) |
C19H34O2 | 294 | Fatty acid ester |
| 44 | 18.764 | 0.97 | 9-Octadecenoic acid, methyl ester, (E)- | C19H36O2 | 296 | Fatty acid ester |
| 45 | 18.869 | 0.05 | Neophytadiene | C20H38 | 278 | Terpenoid |
| 46 | 18.999 | 0.44 | Octadecanoic acid, methyl ester (Methyl stearate) |
C19H38O2 | 298 | Fatty acid ester |
| 47 | 19.066 | 8.9 | 10(E),12(Z)-Conjugated linoleic acid | C18H32O2 | 280 | Fatty acid |
| 48 | 19.118 | 5.36 | Oleic Acid | C18H34O2 | 282 | Fatty acid |
| 49 | 19.324 | 4.29 | Octadecanoic acid (stearic acid) | C18H36O2 | 284 | Fatty acid ester |
| 50 | 19.508 | 0.15 | 9-Octadecenamide | C18H35NO | 281 | Fatty amide |
| 51 | 19.875 | 0.61 | 10(E),12(Z)-Conjugated linoleic acid | C18H35NO | 281 | Fatty amide |
| 52 | 20.202 | 0.13 | Undec-10-ynoic acid, tetradecyl ester |
C25H46O2 | 378 | Fatty acid ester |
| 53 | 21.076 | 0.57 | Eicosanoic acid (Arachidic acid) | C20H40O2 | 312 | Saturated fatty acid |
| 54 | 21.925 | 0.09 | 1H-1,3-Benzimidazol-7-amine, N-[(4-methoxyphenyl)methyl |
C16H17N3O | 267 | Heterocyclic compound |
| 55 | 22.291 | 1.19 | Hexadecanoic acid, 2-hydroxy- 1-(hydroxymethyl)ethyl ester (Glycerol beta-palmitate) |
C19H38O4 | 330 | Fatty acid ester |
| 56 | 22.414 | 0.81 | Tyrosine | C9H11NO3 | 181 | Amino acid |
| 57 | 22.924 | 0.4 | 9-Hexacosene | C26H52 | 364 | Hydrocarbon |
| 58 | 23.005 | 0.23 | 1-Decyloxymethyl-3-methyl-1, 3-dihydrobenzoimidazol-2-ylideneamine |
C19H31N3O | 317 | Heterocyclic compounds |
| 59 | 23.437 | 0.18 | Ethyl 6,9,12,15-octadecatetraenoate | C20H32O2 | 304 | Fatty acid ethyl ester |
| 60 | 23.661 | 0.94 | 9,12-Octadecadienoic acid (Z,Z)-, 2,3-dihydroxypropyl ester |
C21H38O4 | 354 | Fatty acid ester |
| 61 | 23.869 | 0.17 | Octadecanoic acid, 2,3-dihydroxypropyl ester | C21H42O4 | 358 | Fatty acid ester |
| 62 | 23.91 | 0.66 | Benzoic acid, 2,4-dihydroxy-, 4-(1,1-dimethylpropyl)phenyl |
C21H42O4 | 358 | Aromatic ester |
| 63 | 24.308 | 0.16 | 9-octadecenamide (Oleamide) | C18H35NO | 281 | Fatty amide |
| 64 | 24.544 | 0.19 | Squalene | C30H50 | 410 | Triterpenoid |
| 65 | 25.135 | 0.98 | 1,54-dibromotetrapentacontane | C54H108Br2 | 914 | Hydrocarbon |
| 66 | 26.347 | 0.56 | γ-Tocopherol (Vitamin E) | C28H48O2 | 416 | Phenolic compound |
| 67 | 26.659 | 0.77 | Cholesta-4,6-dien-3-ol, (3.beta.) | C27H44O | 384 | Cholesterol |
| 68 | 26.758 | 1.2 | 1-Heptacosanol | C27H56O | 396 | Fatty alcohol |
| 69 | 26.857 | 0.4 | Stigmast-5-en-3-ol, oleat | C47H82O2 | 678 | Phytosterol |
| 70 | 27.055 | 0.49 | α -Tocopherol (Vitamin E) | C29H50O2 | 430 | Phenolic compound |
| 71 | 27.39 | 0.17 | Dodecane, 1,1-dimethoxy- | C14H30O2 | 230 | Acetal |
| 72 | 27.744 | 0.27 | Tetracosan-10-yl acetate | C26H52O2 | 396 | Fatty acid ester |
| 73 | 28.224 | 0.22 | Ergost-5-en-3-ol (3.beta.,24R)- (Campesterol) |
C28H48O | 400 | Phytosterol |
| 74 | 28.509 | 1.91 | Stigmasterol | C29H48O | 412 | Phytosterol |
| 75 | 28.662 | 0.35 | Stigmasta-4,22-diene | C29H48 | 396 | Phytosterol |
| 76 | 28.925 | 0.99 | 1-Hexacosanol (Ceryl alcohol) | C26H54O | 382 | Primary fatty alcohol |
| 77 | 29.266 | 10.84 | Stigmast-5-en-3-ol, (3beta), (β-sitosterol) |
C29H50O | 414 | Phytosterol |
| 78 | 29.431 | 3.82 | Stigmastanol | C29H52O | 416 | Phytosterol |
| 79 | 29.918 | 0.7 | α-amyrin | C30H50O | 426 | Triterpenoid |
| 80 | 30.154 | 1.44 | Lup-20(29)-en-3-one (Lupenone) | C30H48O | 424 | Triterpenoid |
| 81 | 30.58 | 5.09 | Lupeol | C30H50O | 426 | Triterpenoid |
| 82 | 31.12 | 1.85 | γ Sitostenone | C29H48O | 412 | Phytosterol |
| 83 | 32.737 | 1.45 | Cyclopentanone, 2-(5-oxohexyl) | C11H18O2 | 182 | Fatty acid derivative |
| 84 | 33.938 | 0.94 | Stigmastane-3,6-dione, (5-alpha) | C29H48O2 | 428 | Phytosterol |
| R. time: Retention time and NR: Not reported | ||||||
| Table 3: | List of phyto-constituents present in green fruits of C. lancifolius analyzed through GC-MS | |||
| Peak | R. Time | Area % | Name of the compound | Molecular formula |
Molecular weight (g/mol) |
Name of compound |
| 1 | 4.803 | 0.46 | 4H-Pyran-4-one, 2, 3-dihydro-3,5-dihydroxy-6-methyl |
C6H8O4 | 144 | Phenolic |
| 2 | 5.913 | 0.12 | Furaneol | C6H8O3 | 128 | Acetate ester |
| 3 | 6.247 | 0.37 | 1,3,5-Triazine-2,4,6-triamine (Melamine) | C3H6N6 | 126 | Heterocyclic compound |
| 4 | 6.647 | 0.21 | 3,7-Dimethylocta-1,6-dien-3-ol (Linalool) | C10H18O | 154 | Monoterpene alcohol |
| 5 | 7.173 | 0.19 | Pentanoic acid (Valeric acid) | C18H16O2 | 144 | Alkyl carboxylic acid |
| 6 | 7.358 | 2.65 | 4H-Pyran-4-one, 2,3-dihydro- 3,5-dihydroxy-6-methyl |
C6H8O4 | 144 | Phenolic compound |
| 7 | 7.586 | 0.03 | 2-Cyclopenten-1-one, 2-hydroxy- 3-methyl-(Corylon) |
C6H8O2 | 112 | Fatty acid derivatives |
| 8 | 8.604 | 0.19 | 5-Hydroxymethylfurfural | C6H6O3 | 126 | Heterocyclic furan derivative |
| 9 | 8.815 | 0.07 | Butanoic acid, 3-hydroxy-3-methyl | C5H10O3 | 118 | Hydrocarboxylic acid |
| 10 | 9.113 | 0.15 | Decanoic acid (Capric acid) | C10H20O2 | 172 | Fatty acid |
| 11 | 9.337 | 0.05 | Butanamide, 2-hydroxy-N,2,3,3-tetramethyl- | C8H17NO2 | 159 | Branched hydroxyamide |
| 12 | 9.536 | 0.79 | Phenol, 2-methyl-5-(1-methylethyl) | C10H14O | 150 | Phenolic compound |
| 13 | 9.667 | 0.25 | Phenol, 2-methyl-5-(1-methylethyl) | C10H14O | 150 | Phenolic compound |
| 14 | 9.827 | 0.13 | 3-Methylpiperidin-4-ol | C6H13NO | 115 | Heterocyclic alcohol |
| 15 | 10.426 | 0.16 | Phenol, 2-methoxy-4-(2-propenyl) (1,3,4-Eugenol) |
C10H12O2 | 164 | Phenolic compound |
| 16 | 10.623 | 0.48 | 4-tert-Butylcyclohexyl acetate | C12H22O2 | 198 | Carboxylic ester |
| 17 | 10.708 | 26.13 | 1,2,3-benzenetriol (Pyrogallol) | C6H6O3 | 126 | Phenolic compound |
| 18 | 11.791 | 1.78 | Silane, dimethyl(but-2-enyloxy) isobutoxy | C10H22OSi | 202 | Alkoxysilane derivative |
| 19 | 11.994 | 0.97 | 2-Oxovaleric acid, tert-butyldimethylsilyl ester | C11H22O3Si | 230 | Alpha keto acid |
| 20 | 12.25 | 0.24 | Anhydro-d-mannosan (Levoglucosan) | C6H10O5 | 162 | Carbohydrate derivative |
| 21 | 12.683 | 0.72 | 10,12-Tricosadiynoic acid | C23H38O2 | 346 | Fatty acid |
| 22 | 12.868 | 0.21 | 2-[Di(tert-butyl) silyl oxy methyl] tetrahydro furane |
C13H28O2Si | 244 | Silyl ether |
| 23 | 13.019 | 0.32 | Ethyl iso-allocholate | C26H44O5 | 436 | Steroid derivative |
| 24 | 14.128 | 0.36 | Isocitronellol | C10H20O | 156.27 | Monoterpenoid |
| 25 | 14.978 | 0.24 | Caryophyllane, 4,8- beta-epoxy | C15H26O | 222 | Sesquiterpene |
| 26 | 15.311 | 0.66 | Tetradecanoic acid (Myristic acid) | C14H28O2 | 228 | Fatty acid |
| 27 | 15.802 | 1.4 | 1-Heptatriacotanol | C37H76O | 537 | Fatty alcohol |
| 28 | 16.165 | 0.83 | Neophytadiene | C20H38 | 278 | Terpenoid |
| 29 | 16.259 | 0.42 | (E)-3-Methyl-5-((1R,4aR,8aR)-5,5,8a-trimethyl -2-methylenedecahydronaphthalen- 1-yl)pent-2-en-1-ol (Copalol) |
C20H34O | 290 | Diterpenoid |
| 30 | 16.369 | 1.99 | Copalol | C20H34O | 290 | Diterpenoid |
| 31 | 16.42 | 0.29 | 5-isopropyl-6,6-dimethylhept-3-yne-2,5-diol | C12H22O2 | 198 | Oxygenated monoterpoid |
| 32 | 16.62 | 0.8 | Phytol (3,7,11,15-Tetramethyl- 2-hexadecen-1-ol) |
C20H40O | 296 | Diterpenoid |
| 33 | 17.063 | 0.82 | Hexadecanoic acid, methyl ester | C17H34O2 | 270 | Fatty acid ester |
| 34 | 17.413 | 11.12 | n-Hexadecanoic acid (Palmitic acid) | C16H32O2 | 256 | Fatty acid ester |
| 35 | 17.817 | 0.1 | Nonadecane | C19H40 | 268 | Alkane |
| 36 | 18.067 | 0.18 | 7-Hexadecenoic acid, methyl ester, (Z)- | C17H32O2 | 268 | Fatty acid ester |
| 37 | 18.375 | 0.21 | 9-octadecenoic acid (z) | C19H36O2 | 296 | Fatty acid ester |
| 38 | 18.702 | 0.86 | 9,12-Octadecadienoic acid (Z, Z), methyl ester (Methyl linoleate) |
C19H34O2 | 294 | Fatty acid ester |
| 39 | 18.764 | 0.75 | 9-Octadecenoic acid, methyl ester, (E) | C19H36O2 | 296 | Fatty acid ester |
| 40 | 18.869 | 0.14 | Phytol (3,7,11,15-Tetramethyl- 2-hexadecen-1-ol) |
C20H40O | 296 | Diterpenoid |
| 41 | 19.053 | 2.66 | 10(E),12(Z)-Conjugated linoleic acid | C18H32O2 | 280 | Fatty acid |
| 42 | 19.109 | 2.85 | cis-9-Hexadecenal | C16H30O | 238 | Unsaturated aldehyde |
| 43 | 19.317 | 2.46 | Octadecanoic acid | C18H36O2 | 284 | Fatty acid ester |
| 44 | 19.562 | 0.97 | 9(E),11(E)-Conjugated linoleic acid (ethyl linoleate) |
C18H32O2 | 280 | Fatty acid ethyl ester |
| 45 | 20.51 | 0.07 | Myristic acid glycidyl ester | C17H32O3 | 284 | Fatty acid ester |
| 46 | 20.731 | 0.1 | 7-Hexadecenal, (Z)- | C16H30O2 | 238 | Unsaturated aldehyde |
| 47 | 21.077 | 0.5 | 9-Octadecenoic acid (Z)- | C19H36O2 | 296 | Fatty acid ester |
| 48 | 21.345 | 0.14 | Behenic alcohol | C22H46O | 326 | Fatty alcohol |
| 49 | 22.192 | 0.13 | Heneicosane | C21H44 | 296 | Hydrocarbon |
| 50 | 22.29 | 1.26 | Hexadecanoic acid, 2-hydroxy- 1-(hydroxymethyl)ethyl ester |
C19H38O4 | 330 | Fatty acid ester |
| 51 | 22.578 | 0.22 | Docosyl pentafluoro propionate | C25H45F5O2 | 472 | Long-chain fluorinated ester |
| 52 | 22.72 | 0.1 | 1-octanol, 3,7-dimethyl | C10H22O | 158 | Fatty acid ester |
| 53 | 22.894 | 0.03 | 3-Methylbutylhexa decanoate | C21H42O2 | 326 | Fatty acid ester |
| 54 | 23.662 | 0.24 | 6,9- Octadecadienoic acid, methyl ester | C19H34O2 | 294 | Fatty acid ester |
| 55 | 23.708 | 0.16 | 2-Methylhexacosane | C27H56 | 380 | Hydrocarbon |
| 56 | 23.867 | 0.24 | Octadecanoic acid, 2,3-dihydroxypropyl ester | C23H42O4 | 358 | Fatty acid ester |
| 57 | 24.31 | 0.27 | 13-Docosenamide, (Z) (Erucamide) | C22H43NO | 337 | Fatty acid amide |
| 58 | 25.143 | 1.95 | 1-Heptacosanol | C27H56O | 396 | Fatty alcohol |
| 59 | 25.899 | 0.29 | Heptacosyl heptafluorobutyrate | C31H55F7O2 | 592 | Hydrocarbon |
| 60 | 26.344 | 0.31 | γ -Tocopherol | C28H48O2 | 416 | Phenolic compound |
| 61 | 26.66 | 0.3 | Cholesta-4,6-dien-3-ol, (3-beta) | C27H44O | 384 | Cholesterol |
| 62 | 26.757 | 4.43 | 1-Heptacosanol | C27H56O | 396 | Fatty alcohol |
| 63 | 27.049 | 0.22 | α-Tocopherol | C29H50O2 | 430 | Phenolic compound |
| 64 | 28.501 | 1.04 | Stigmasta-5,23-dien-3-ol, (3-beta), (Stigmasterol) |
C29H48O | 412 | Phytosterol |
| 65 | 28.924 | 1.14 | 1-Hexacosanol (Ceryl alcohol) | C26H54O | 382 | Primary fatty alcohol |
| 66 | 29.259 | 9.57 | β - Sitosterol | C29H50O | 414 | Phytosterol |
| 67 | 29.432 | 2.1 | Stigmastanol | C29H52O | 416 | Phytosterol |
| 68 | 29.919 | 0.37 | β-Amyrin | C30H50O | 426 | Triterpenoid |
| 69 | 30.158 | 1.31 | Lup-20(29)-en-3-one (Lupenone) | C30H48O | 424 | Triterpenoid |
| 70 | 30.579 | 5.57 | Lupeol | C30H50O | 426 | Triterpenoid |
| 71 | 31.116 | 1.15 | Gamma-sitostenone | C29H48O | 412 | Phytosterol |
| R. time: Retention time and NR: Not reported | ||||||
| Table 4: | Phytochemical composition in methanolic extract of dry (Dfr) and green fruits (Gfr) of C. lancifolius | |||
| Phytochemical group | Total % area in Dfr | Total % area in Gfr |
| Fatty acid derivatives | 38.09 | 39.43 |
| Terpenes, terpenoids and derivatives | 19.04 | 16.9 |
| Phenolic compounds and derivatives | 9.52 | 11.26 |
| Phytosterols | 9.52 | 5.63 |
| Hydrocarbons | 5.95 | 8.45 |
| Heterocyclic compounds | 5.95 | 4.22 |
| Amino acid | 1.19 | - |
| Miscellaneous compounds | 10.71 | 14.08 |
The unique compounds which were observed in green fruits as not observed in dry fruits of C. lancifolius are β-Amyrin, Heptacosyl heptafluorobutyrate, Erucamide, 2-Methylhexacosane, Docosyl pentafluoro propionate, 1-octanol, 3,7-dimethyl, 3-Methylbutylhexadecanoate, 6,9- Octadecadienoic acid, methyl ester, 2-Methylhexacosane, Octadecanoic acid, 2,3-dihydroxypropyl ester, 9-Octadecenoic acid (Z)-, Behenic alcohol, Heneicosane, 9(E),11(E)-Conjugated linoleic acid, Myristic acid glycidyl ester, 7-Hexadecenal, (Z)-, cis-9-Hexadecenal, 7-Hexadecenoic acid, methyl ester, (Z)-, Nonadecane, Copalol, 5-isopropyl-6,6-dimethylhept-3-yne-2,5-diol, 1-Heptatriacotanol, Ethyl iso-allocholate, 2-[Di(tert-butyl) silyl oxy methyl] tetrahydro furan, Silane, dimethyl(but-2-enyloxy) isobutoxy, 2-Oxovaleric acid, tert-butyldimethylsilyl ester, Levoglucosan, 3-Methylpiperidin-4-ol, Butanamide, 2-hydroxy-N,2,3,3-tetramethyl-, Butanoic acid, 3-hydroxy-3-methyl, 5-Hydroxymethylfurfural, Corylon, Pentanoic acid, Furaneol, Melamine and Linalool. Whereas the 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl, Phytol, Phenol, 2-methyl-5-(1-methylethyl), Copalol, 1-Heptacosanol and 9-Octadecenoic acid (Z)- gave double peaks (Table 3).
DISCUSSION
Due to the ecological and therapeutic importance, research on secondary metabolites has become a key focus in fields such as organic chemistry, pharmacology, molecular biology, and bioinformatics16. GC-MS is a method that integrates the separation of phytochemicals using gas chromatography with their detection via mass spectrometry and is mostly utilized for identifying and characterizing volatile and semi-volatile components in intricate plant matrices. For phytochemical investigations, the methanolic extracts are mostly used because methanol is an effective solvent for dissolving a variety of polar and moderately non-polar compounds3. Hence, in the present investigation, methanol was utilized for the extraction of maximum phyto-constituents.
The constituents of the methanolic extract of C. lancifolius were classified into major phytochemical groups based on their structural classes as determined by GC-MS analysis (Table 4). Many of these compounds have recognized pharmaceutical significance, with previous studies reporting diverse biological activities, like antioxidant, antidiabetic, anticancer, analgesic, antimicrobial, and anti-inflammatory etc. (Table 2 and 3). Fatty acid derivatives constituted the predominant chemical class, accounting for 38.09% and 39.43% of the total composition in Dfr and Gfr, respectively (Table 4). This predominance primarily resulted from the high abundance of compounds such as palmitic acid, myristic acid, 10(E),12(Z)-conjugated linoleic acid, oleic acid, stearic acid, capric acid, lauric acid, as well as 9-octadecenoic acid methyl ester (E) in Dfr (Table 2) and hexadecanoic acid, 1-heptacosanol, stearic acid, ceryl alcohol, 9-octadecenoic acid methyl ester (E), and cis-9-hexadecenal in Gfr (Table 3).
Fatty acids represent one of the most fundamental classes of biomolecules, exhibiting diverse biological activities with notable therapeutic relevance. Terpenoids, the second most abundant class of biomolecules in the present study (Table 4), are multifunctional secondary metabolites structurally derived from isoprene (C5) units in plants. C. lancifolius fruits were also found to possess a considerable number of phenolic compounds which have a benzene ring with one or more hydroxyl groups and can occur in a variety of structures such as phenylpropanoids, flavonoids, tannins, melanins, lignins, etc. On the other hand, phytosterols are natural triterpenoids in plants having a tetracyclic structure with functional groups usually at C-3,4,7,12,17 positions. Hydrocarbons are organic compounds made up of carbon and hydrogen atoms and classified as aliphatic/alicyclic or aromatic compounds. Remarkably, all of these secondary metabolites have shown to possess various biological activities for example, antioxidant, antimicrobial, anti-inflammatory, anticancer, antiangiogenic, antidiabetic, hypolipidemic, neuroprotective, immunomodulatory, hepatoprotective etc.1,17,18. Thus, the rich phytochemical profile of C. lancifolius fruits could motivate pharmacologists for novel drug discovery.
Bonnet et al.19 have shown that green (immature) fruits of Musa acuminata contained more polyphenol content. Similar results were also observed in this study, where green fruits of C. lancifolius showed more phenolic compounds (11.26%) than dry mature fruits (9.52%). Likewise, higher terpenoid and phytosterol contents were observed in mature fruits of C. lancifolius (28.56%) than in its green fruits (22.53%), as shown in Table 4. These results are similar to a study by Simchuer and Srihanam20where higher triterpenoids and sterol contents were observed in ripe fruits of Ampelocissus martini than in immature fruits. Moreover, high concentrations of β-sitosterol were also found in ripe fruits, comparable to the findings of the present study, where 10.84% β-sitosterol is found to be present in dry mature fruits and 9.57% is found in green fruits.
The compounds detected in the present study have also been reported in other plant parts of C. lancifolius. For example, Al-Shatti et al.21 reported 4.90% pyrogallol, 5.61% heneicosane and 5.61% dianhydromannitol from leaves of C. lancifolius. Moni et al.22 also reported various bioactive compounds from hot methanolic extract of the leaves of C. lancifolius through GC-MS, some of which are similar to the present study, such as phytol, hexadecanoic acid, campesterol and oleic acid. The essential oil obtained from leaves of C. lancifolius revealed presence of 85 compounds through GC-MS among which the common compounds in both leaves and fruits are Hexadecanoic acid, Henicosane, Hexacosane, Squalene, γ-Sitosterol, Lupenone, Lupeol, Eicosanoic acid, Neophytadiene, Tetradecanoic acid and Dodecanoic acid with relative abundance % of 2.97, 0.29, 3.06, 1.09, 0.07, 0.61, 3.29, 0.08, 0.26, 0.48, 0.45, respectively as reported by Salim et al.14.
Interestingly, several phytochemicals detected in the current study have been previously reported in some other plant species of the Combretaceae family, though their relative abundance was variable for example, 4.17% 1-Heptacosanol, 0.5% Hexadecanal, 10.48% γ-Sitosterol were reported in the root of Terminalia travancorensis23. Recently, Sarvendra et al.24reported various similar phyto-compounds such as 2.66% Phytol, 5.88% Palmitic acid, 2.61% Neophytadiene, 0.12% Heptadecanoic acid, 2.62% Linoleic acid, 0.44% Behenic acid, 1.17% Heptacosane, and 2.31% Squalene from leaves of Combretum indicum. The difference in concentration of these phyto-constituents might be the result of variations at the generic level, edaphic, climatic, and geographical factors, as well as conditions of plant collection and extraction.
Besides, some of these compounds are allelopathic in nature, for example, hexacosane and hexadecanoic acid ethyl ester, which are also found in the leaves of an invasive alien plant species, Lantana camara4. This needs to be investigated in detail as C. lancifolius is also an exotic species for India, and questions related to its suitability for plantation in various Indian states are being raised. Therefore, the results of this study may stimulate the assessment of the dual biological roles, i.e., ecological impact and therapeutic potential of C. lancifolius.
CONCLUSION
GC-MS analysis of the methanolic extract of Conocarpus lancifolius fruits unveiled a wide spectrum of bioactive phytochemicals, which have promising therapeutic potential. The major metabolites, particularly lupeol, β-sitosterol acetate, stigmastanol, pyrogallol, linoleic acid, and phytol, dominate the profile and also possess antioxidant, antimicrobial, anti-inflammatory, and lipid-lowering activities. This is the first GC-MS-based comparative study of mature and green (immature) fruits of C. lancifolius and lays the foundation for further bioassay-guided isolation, toxicity assessment, and pharmacological evaluation to find novel drugs.
SIGNIFICANCE STATEMENT
This study provides the first comprehensive evaluation of phyto-constituents from the methanolic extracts of both mature and immature fruits of Conocarpus lancifolius through the GC-MS technique. These phytochemicals are reported to possess a wide range of pharmacological activities. Hence, the present investigation will pave the way for a detailed evaluation of the therapeutic potential of its fruits. Moreover, this study will encourage the researchers to further investigate the allelopathic nature of these compounds, since C. lancifolius is an exotic plant species for India and may become an Invasive Alien Plant Species in the future.
ACKNOWLEDGMENTS
Authors are thankful to the Advanced Instrumentation Research Facility (AIRF), Jawahar Lal Nehru University, New Delhi, India, for providing the GC-MS facility. First author is also thankful to CSIR-UGC, New Delhi, for providing financial assistance in the form of Junior Research Fellowship.
REFERENCES
- Palazon, J. and M.A. Alcalde, 2025. Secondary metabolites in plants. Plants, 14.
- Mohamed, A.A., A.A. Khalil and H.E.S. El-Beltagi, 2010. Antioxidant and antimicrobial properties of kaff maryam (Anastatica hierochuntica) and doum palm (Hyphaene thebaica). Grasas Aceites, 61: 67-75.
- Kumari, A. and V. Jain, 2025. Phytochemical composition and GC-MS analysis of Hymenodictyon orixense Mabb. leaves. GSC Biol. Pharm. Sci., 32: 156-179.
- Kayesth, S. and K.K. Gupta, 2018. Impact of Lantana camara hexane extract on survival, growth and development of Dysdercus koenigii Fabricius (Heteroptera: Pyrrhocoriedae). Acta Ecol. Sinica, 38: 187-192.
- Redha, A., R. Al-Hasan and M. Afzal, 2021. Synergistic and concentration-dependent toxicity of multiple heavy metals compared with single heavy metals in Conocarpus lancifolius. Environ. Sci. Pollut. Res., 28: 23258-23272.
- Al-Taweel, A.M., S. Perveen, G.A. Fawzy, R. Mehmood, A. Khan and S.I. Khan, 2016. New ellagic acid derivative from the fruits of heat-tolerant plant Conocarpus lancifolius Engl. and their anti-inflammatory, cytotoxic, PPAR agonistic activities. Pak. J. Pharm. Sci., 29: 1833-1837.
- Afifi, H.S., H.M. Al Marzooqi, M.J. Tabbaa and A.A. Arran, 2021. Phytochemicals of Conocarpus spp. as a natural and safe source of phenolic compounds and antioxidants. Molecules, 26.
- Zaman, K., F. Rahim, M. Taha, M. Sajid and S. Hayat, 2021. Synthesis, in vitro antiurease, in vivo antinematodal activity of quinoline analogs and their in-silico study. Bioorg. Chem., 115.
- Abdel Bar, F.M., A.A. Salkini, Y. Amen and A.E. Sherif, 2023. Acetylcholinesterase inhibitors from Conocarpus lancifolius Engl. (Combretaceae). Nat. Prod. Res., 37: 1668-1673.
- Saadullah, M., A. Farid, A. Ali, M. Rashad and F. Naseem et al., 2022. Molecular modeling study of novel Lancifolamide bioactive molecule as an inhibitor of acetylcholinesterase (AChE), herpes simplex virus (HSV-1), and anti-proliferative proteins. Molecules, 27.
- Khurm, M., Y. Guo, Q. Wu, X. Zhang and M.U. Ghori et al., 2023. Conocarpus lancifolius (Combretaceae): Pharmacological effects, LC-ESI-MS/MS profiling and in silico attributes. Metabolites, 13.
- Moglad, E., S. El-Shae, M. Allam and H.A. Algahtan, 2023. Antimicrobial and antiquorum-sensing activity of Conocarpus lancifolius Engl. (Combretaceae). Emir. J. Food Agric., 35: 197-202.
- Prajapati, P., B.B. Maitreya and R.M. Rawal, 2024. Qualitative and quantitative phytochemical screening and chemical fingerprint analysis of Conocarpus lancifolius plant using HPTLC. Vegetos, 38: 1506-1514.
- Salim, A., A.A. Arasteh, R. Sahrish, D. Labash, A.A. El-Keblawy, H.A. Gad and N.S. Ashmawy, 2025. Comparative metabolic profiling and biological evaluation of essential oils from Conocarpus species: antidiabetic, antioxidant, and antimicrobial potential. Plants, 14.
- Kunwar, B., V. Jain and S.K. Verma, 2024. In vitro thrombolytic potential of a nutritive vegetable-Momordica dioica Roxb. ex Willd. Asian J. Biol. Sci., 17: 228-234.
- Lal, N., N. Sahu, A.O. Shirale, P. Gurav and K. Rani et al., 2023. Plant Secondary Metabolites and their Impact on Human Health. In: Nano-Biofortification for Human and Environmental Health, Rajput, V.D., H. El-Ramady, S.K. Upadhyay, T. Minkina, B. Ahmed and S. Mandzhieva, Springer International Publishing, Switzerland, ISBN: 978-3-031-35147-1, pp: 295-321.
- Russo, M., L. Spagnuolo, G. Cafeo and L. Dugo, 2025. Phenolic Compounds (Extraction, Quantification, Biological Activities). In: Phytochemicals for Health, Locatelli, M., M. Tomczyk, L. Dugo and M. Russo, Elsevier, Amsterdam, Netherlands, ISBN: 978-0-443-15366-2, pp: 397-420.
- Begum, I.F., R. Mohankumar, M. Jeevan and K. Ramani, 2016. GC-MS analysis of bio-active molecules derived from Paracoccus pantotrophus FMR19 and the antimicrobial activity against bacterial pathogens and MDROs. Indian J. Microbiol., 56: 426-432.
- Bonnet, C.B., O. Hubert, D. Mbeguie-A-Mbeguie, D. Pallet, A. Hiol, M. Reynes and P. Poucheret, 2013. Effect of physiological harvest stages on the composition of bioactive compounds in Cavendish bananas. J. Zhejiang Univ.-Sci. B, 14: 270-278.
- Simchuer, W. and P. Srihanam, 2018. Phytosterol screening of skin and seed extracts of wild grape Ampelocissus martinii Planch. fruits. Orient. J. Chem., 34: 875-880.
- Al-Shatti, A.H., A. Redha, P. Suleman and R. Al-Hasan, 2014. The allelopathic potential of Conocarpus lancifolius (Engl.) leaves on dicot (Vigna sinensis L.), monocot (Zea mays L.) and soil-borne pathogenic fungi. Am. J. Plant Sci., 05: 2889-2903.
- Moni, S.S., M.F. Alam, M.H. Sultan, H.A. Makeen and H.A. Alhazmi et al., 2021. Spectral analysis, in vitro cytotoxicity and antibacterial studies of bioactive principles from the leaves of Conocarpus lancifolius, a common tree of Jazan, Saudi Arabia. Braz. J. Biol., 83.
- Lakshmi, M. and B.R. Nair, 2017. GC-MS analysis of the chloroform extract of bark of Terminalia travancorensis Wight & Arn. (Combretaceae). Int. J. Pharm. Sci. Res., 8: 794-798.
- Sarvendra, K., S. Chaubey, V.K. Shukla, A. Patel and S.C. Sati, 2025. Comprehensive GC-MS profiling of methanolic and chloroform extracts of Combretum indicum leaf for therapeutic potential use. YMER, 24: 26-35.
How to Cite this paper?
APA-7 Style
Mittal,
V., Jain,
V. (2026). Phytochemical Profiling and GC-MS Analysis of Conocarpus lancifolius Fruits. Trends in Biological Sciences, 2(1), 87-98. https://doi.org/10.21124/tbs.2026.87.98
ACS Style
Mittal,
V.; Jain,
V. Phytochemical Profiling and GC-MS Analysis of Conocarpus lancifolius Fruits. Trends Biol. Sci 2026, 2, 87-98. https://doi.org/10.21124/tbs.2026.87.98
AMA Style
Mittal
V, Jain
V. Phytochemical Profiling and GC-MS Analysis of Conocarpus lancifolius Fruits. Trends in Biological Sciences. 2026; 2(1): 87-98. https://doi.org/10.21124/tbs.2026.87.98
Chicago/Turabian Style
Mittal, Vaishali, and Vartika Jain.
2026. "Phytochemical Profiling and GC-MS Analysis of Conocarpus lancifolius Fruits" Trends in Biological Sciences 2, no. 1: 87-98. https://doi.org/10.21124/tbs.2026.87.98

This work is licensed under a Creative Commons Attribution 4.0 International License.




