Antivenom Potentials of Local Medicinal Plants Against Echis ocellatus Envenomation in Aliero, Kebbi State, Nigeria
| Received 29 Oct, 2025 |
Accepted 07 Mar, 2026 |
Published 30 Jun, 2026 |
Background and Objective: Snakebite injuries, majority of whom are people who live in remote, poorly developed, and underdeveloped or developing tropical regions of the world. The venom of Echis ocellatus is highly toxic, primarily comprising majorly of metalloproteinases which possess hemorrhagic, nephrotoxic, cardiotoxic, and anti-coagulant effects. This research work aimed to assess the antivenom potentials of local medicinal plants found in Aliero, Kebbi State, Nigeria, against the venom of E. ocellatus. Materials and Methods: A snake Charmer was used to capture the E. ocellatus and duly identified by a Zoologist. The snake venom was milked, and its lethal doses were evaluated by probit analysis. The medicinal plants were collected, and their extracts were obtained using methanol. The crude methanol extracts were screened for antivenom activity using an albino rat model. The data are expressed as Mean±SEM and analyzed by one-way ANOVA with differences separated using Duncan’s Multiple Range Test (p<0.05) via IBM SPSS Statistics 20. Results: The lethal doses, 50% (LD50) and 100% (LD100) of E. ocellatus venom, were determined to be 0.316 mg/kg body weight and 3.55 mg/kg b. wt. respectively. The antivenom activity of some medicinal plants against E. ocellatus revealed non-significant (p>0.05) differences in mean survival time of Sclerocarya birrea (A.Rich.) Hochst leaves, Catunaregam nilotica (Stapf) Tirveng root and Crinum ornatum(Aiton) Herb. bud,Sclerocarya birrea (A.Rich.) Hochst root, Ficus platyphyllaDelile stembark andFaidherbia albida(Delile) A.Chev root methanol extracts compared to both normal and positive control. While M. inermisroot revealed a significant (p<0.05) decrease in mean survival time compared to both normal and positive control, and was not significantly (p>0.05) different compared to negative control. Conclusion: The findings of this study document the lethal profile of Echis ocellatusvenom and further disclose that some indigenous medicinal plants used in Aliero, Kebbi State, have potent antivenom potentials and thus can serve as a guide towards the synthesis and development of conventional antivenoms.
| Copyright © 2026 Sani et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Snakebite injuries affect an estimated 2.7 million people yearly; many of them are living in isolated, undeveloped tropical areas of the world1. Annual snakebite cases range between 81,000 to 138,000 in Africa, while more than 400,000 cases lead to permanent physical and mental disabilities2. Snakebite envenoming is considered to be a neglected tropical disease3. This incidence is increasing mostly in rural communities and some parts of urban areas4. The spike in incidence of snake bites might be attributed to the daily activities of these people, most especially in Northern Nigeria, where the majority of these people are farmers and animal rearing, many of these people are less privileged with poor shelter and thus are at risk of snake attack even at their comfort zones5.
In West Africa, Carpet viper (Echis ocellatus) is considered to be one of the medically snake species6. Its bite is highly poisonous and lethal, with a lethality index higher than other species in Africa combined. Echis ocellatus venom is very toxic, that, mainly comprising metalloproteinases, causing haemorrhage, cardio- and nephro-toxicity as well as anticoagulant activity7. Hence, E. ocellatus bite results in numerous clinical complications ranging from local to severe systemic damages like inflammation, blistering, haemorrhages and necrosis7.
Several efforts were made by researchers in developing effective snakebite therapies, but, still, conventional antivenoms remain the only solution8. There are many limitations with the synthetic antivenoms, ranging from serum sickness, anaphylactic reactions, to inability in preventing local tissue damage after snakebite9. The conventional antivenoms, apart from causing adverse effects, are mostly not affordable and available in rural communities in which snakebite cases are common10.
There were encouraging positive outcomes from the screening of many species of local medicinal plants against snakebite. This is attributed to their content of many pharmacologically potent phytochemicals. This resulted in their incorporation into numerous conventional therapies11. Hence, this study aims to document the lethality and validity of Echis ocellatus venom and antivenom activities of some local medicinal plants, respectively.
MATERIALS AND METHODS
Ethical statement: All animal experiments were conducted in accordance with the National as well as Kebbi State University of Science and Technology, Aliero Research Ethics Committee guidelines for the use and care of experimental animals, with Ref. No.: KSUSTA/DVC-R&I/RECC/003.
Study area: The research was conducted within Aliero town, Nigeria. It was performed in the Biochemistry Research Laboratory, Department of Biochemistry, Faculty of Life Sciences, Abdullahi Fodio University of Science and Technology, Aliero, Nigeria, between July, 2025 to October, 2025.
Experimental animals: Adult Wister albino rats of both sexes, aged 4-5 months and weighing between 120 and 150 g, were used for the experiments. They were purchased from the National Veterinary Research Institute, Vom, Nigeria, and kept under standard laboratory conditions (22-24°C; 12:12 hrs dark/light cycle). The animals were allowed free access to both food (commercial rodent pellets) and water ad libitum; they were allowed to acclimatize for 2 weeks. The weight of each rat was taken before the commencement of the experiment. All animal experiments were conducted in accordance with the guidelines for the use and care of experimental animals12.
Standard snake venom antiserum (antivenin): The lyophilized polyvalent snake venom antiserum (Batch No.: 8904012480039, Manufacture Date: November 2022, Expiry Date: October 2026) was used as a standard to compare with the efficacys of the plant extract. It was produced by a standard pharmaceutical company (Bharat Serums and Vaccines Limited, India).
Echis ocellatus: The snake species (E. ocellatus) used was captured and housed in a wooden cage with the help of a snake charmer. After collection, it was duly identified by a zoologist in the Department of Animal and Environmental Biology, Kebbi State University of Science and Technology, Aliero, Nigeria. Its venom was milked and used for the experiments.
Milking of venom: The venom was collected between the hours of 17.30-18.00, in a low light condition at ambient temperature according to the method of Goswami et al.13 by using a short-acting general anesthesia; halothane (Piramal Healthcare Limited, U.K.). The glands below the eyes of the snake were compressed to release the stored venom into a clean and sterilized container.
Preparation of venom: After milking, the venom was lyophilized using a freeze-dryer (Mill rock Technology, USA) and kept inside a refrigerator (HR135A, Haier-Thermocool, Lagoss, Nigeria) in a light-resistant and air-tight container. Before use, the lyophilized venom was reconstituted in 0.9 % saline (regarded as the venom) and kept at 4°C. The venom concentration was expressed in terms of dry weight (mg/mL)14.
Determination of venom lethal doses, 50% (LD50) and 100% (LD100): The lethal doses of the venom were determined using a modified method of Theakston and Reid15. Twenty (20) rats were randomly distributed into five (5) groups of four (4) rats each. The venom was reconstituted in normal saline and was administered intraperitoneally (IP) as follows:
| • | Group 1: Served as normal control and were administered with normal saline (i.p.) | |
| • | Group 2: Were injected (i.p.) with the venom at a dose of 1.0 mg/kg b.wt | |
| • | Group 3: Were injected (i.p.) with the venom at a dose of 2.0 mg/kg b.wt | |
| • | Group 4: Were injected (i.p.) with the venom at a dose of 3.0 mg/kg b.wt | |
| • | Group 5: Were injected (i.p.) with the venom at a dose of 4.0 mg/kg b.wt |
Mortality was recorded within 24 hrs of venom administration, and the lethal doses (LD50 and LD100) were estimated using probit analysis15,16. The percentage mortality at zero and 100 was calculated using the formulae:
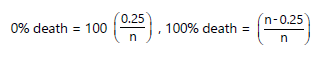 |
Collection and authentication of the plant materials: Mitragyna inermis (Wild) Kuntze Root, Sclerocarya birrea (A.Rich.) Hochst leaves, Sclerocarya birrea (A.Rich) Hochst Root, Ficus platyphylla Delile Stembark, Faidherbia albida (Delile) A. Chev Root, Catunaregam nilotica (Stapf) tirveng Root, and Crinum ornatum bud were collected within Aliero town, Kebbi State, Nigeria. The plants were then authenticated at the herbarium of the Department of Plant Science and Biotechnology, Abdullahi Fodio University of Science and Technology, Aliero, and voucher specimens for Mitragyna inermis (Wild) Kuntze Root [AFUSTA/PSB/H/VOUCHER NO: S.N], Sclerocarya birrea (A.Rich.) [AFUSTA/PSB/H/VOUCHER NO: 114A], Ficus platyphylla Delile Stembark [AFUSTA/PSB/H/VOUCHER NO: SN], Faidherbia albida (Delile) A. Chev [AFUSTA/PSB/H/VOUCHER NO: 319], Catunaregam nilotica (Stapf) tirveng [AFUSTA/PSB/H/VOUCHER NO: SN] and Crinum ornatum (Aiton) Herb. [AFUSTA/PSB/H/VOUCHER NO: SN] were deposited in the herbarium.
Preparation of plants methanol extracts: The plant extracts were prepared according to a modified method of Dupont et al.17. The collected plant parts were washed with clean water and air-dried under shade, pulverized using a pestle and mortar. One hundred grams (100 g) each of the powdered plant part was measured and soaked in 100 mL of 99% methanol. The mixture was then kept at room temperature for 24 hrs and filtered twice; initially with a muslin cloth and later with a Whatman filter paper No.1. The filtrate was evaporated to dryness at 45°C using a rotary evaporator.
Antivenom activity screening of the plants' methanol extracts: Forty albino rats were randomly distributed into seven 10 groups of five 4 rats each. And venom inducement and extract treatment were conducted as follows:
| Groups | Treatments |
| Group 1: | Received orally with only distilled water and served as a normal control |
| Group 2: | Were injected intraperitoneally (i.p.) only with LD100 of the snake venom and served as venom control |
| Group 3: | Were injected (i.p.) with the LD100 of the snake venom, then after 30 min, they were administered intravenously (i.v.) with the standard conventional serum antivenin at the dose of 1 mL/0.45 mg venom and served as standard control |
| Group 4: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Mitragyna inermis (Wild.) Kuntze Root 300 mg/kg b.wt |
| Group 5: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Sclerocarya birrea (A.Rich.) Hochst leaf 300 mg/kg b.wt |
| Group 6: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Sclerocarya birrea (A.Rich.) Hochst Root 300 mg/kg b.wt |
| Group 7: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Ficus platyphylla Delile Stembark 300 mg/kg b.wt |
| Group 8: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Faidherbia albida (Delile) A.Chev Root 300 mg/kg b.wt |
| Group 9: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Catunaregam nilotica (Stapf) Tirveng root 300 mg/kg b.wt |
| Group 10: | Injected (i.p.) with the LD100 of the snake venom, then after 30 min treated with Crinum ornatum (Aiton) Herb. bud 300 mg/kg b.wt |
All the groups received the same volume of preparations. In all the groups, the duration of survival and the number of rats survived were recorded for 24 hrs18.
Data analysis: The data generated from the study are presented as Mean±SEM and subjected to One-way Analysis of Variance (ANOVA), and statistical differences between the means were separated using New Duncan’s Multiple Range Test at p<0.05 with the aid of a statistical package (IBM SPSS Statistics 20).
RESULTS
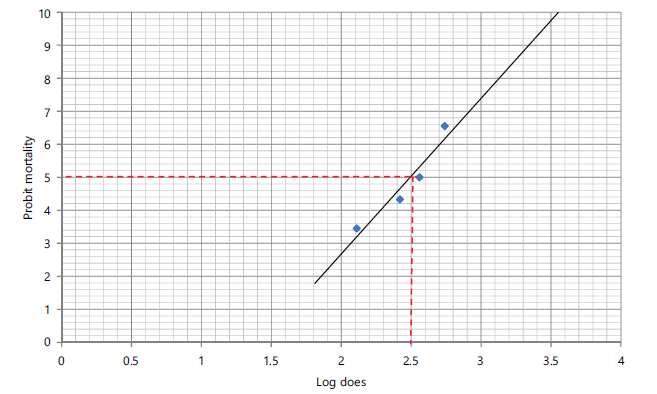
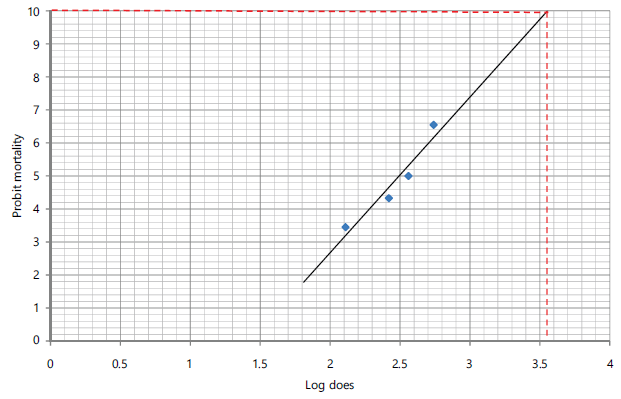
The lethality profile of the Echis ocellatus venom is presented in Table 1. The lethal profile of the E. ocellatus venom showed a clear dose-dependent increase in mortality. No mortality was observed in the control group, and at 1.00 mg/kg, while 25 and 50% mortality were recorded at venom doses of 2.00 and 3.00 mg/kg, respectively. Complete mortality (100%) occurred at 4.00 mg/kg, confirming this dose as lethal. Probit values increased progressively from 3.45 to 6.55 with increasing venom dose, indicating a strong positive relationship between venom concentration and mortality. The LD50 (median lethal dose) and LD100 (maximum lethal dose) of the venom were calculated using probit curves (Fig. 1 and 2, respectively), and were evaluated to be 0.316 mg/kg body weight and 3.55 mg/kg b.wt. respectively.
Neutralization effect of the plant extracts against the Echis ocellatus venom: The neutralization activity of the medicinal plants against the Echis ocellatuss venom is shown in Table 2. The normal control and standard polyvalent anti-venom groups showed 100% survival with a mean survival time of 24.00±0.00 hrs, whereas the negative control showed 0% survival and a significantly reduced survival time (2.30±0.31 hrs). Among the plant extracts, Faidherbia albida and Catunaregam nilotica root methanol extracts provided complete protection (100% survival), comparable to the positive control (p>0.05).
|
|
| Table 1: | Lethal profile of Echis ocellatus venom | |||
| Group | Average animal weight (g) |
Venom dose (mg/kg b.wt) |
Average dose of venom administered (μg/kg b.wt) |
Log dose | No. of death/ No. of rats used |
Death (%) | Corrected* (%) | Probit of Mortality |
| 1 | 153.53 | - | - | - | 0/4 | 0 | 0 | - |
| 2 | 127.57 | 1 | 127.57 | 2.11 | 0/4 | 0 | *6.25 | 3.45 |
| 3 | 131.7 | 2 | 263.44 | 2.42 | 01-Apr | 25 | 25 | 4.33 |
| 4 | 120.64 | 3 | 361.9 2 | 2.56 | 02-Apr | 50 | 50 | 5 |
| 5 | 136.4 | 4 | 545.6 | 2.74 | 04-Apr | 100 | *93.75 | 6.55 |
| Table 2: | Anti-venom activities of the plants extracts against Echis ocellatus venom LD100 | |||
| Groups | Treatment | Treatment (dose) | Survival/ No. of animals used |
Survival (%) | Mean survival time |
| 1 | Normal control | Normal saline (0.5 mL IP) | 04-Apr | 100 | 24.00±0.00c |
| 2 | Negative control | - | 0/4 | 0 | 2.30±0.31a |
| 3 | Positive control (Standard Polyvalent Anti-Venom) |
1 mL/0.6mg Venom | 04-Apr | 100 | 24.00±0.00c |
| 4 | Mitragyna inermis (Wild.) Kuntze Root Methanol Extract |
300 (mg/kg b.wt.) | 01-Apr | 25 | 10.70±4.78ab |
| 5 | Sclerocarya birrea (A.Rich.) Hochst leaves Methanol Extract |
300 (mg/kg b.wt.) | 03-Apr | 75 | 18.99±5.01bc |
| 6 | Sclerocarya birrea (A.Rich.) Hochst Root Methanol Extract |
300 (mg/kg b.wt.) | 02-Apr | 50 | 13.67±5.98bc |
| 7 | Ficus platyphylla Delile Stembark Methanol Extract |
300 (mg/kg b.wt.) | 02-Apr | 50 | 15.97±10.71bc |
| 8 | Faidherbia albida (Delile) A.Chev Root Methanol Extract |
300 (mg/kg b.wt.) | 04-Apr | 100 | 24.00±0.00c |
| 9 | Catunaregam nilotica (Stapf) Tirveng Root Methanol Extract |
300 (mg/kg b.wt.) | 04-Apr | 100 | 24.00±0.00c |
| 10 | Crinum ornatum (Aiton) Herb. Bud Methanol Extract |
300 (mg/kg b.wt.) | 03-Apr | 75 | 19.76±4.24bc |
| Values are presented as Mean±SEM (n = 4). Value having similar alphabetical superscripts are not significantly different at (p>0.05) analyzed using One-Way ANOVA, followed by Duncan multiple comparison test with SPSS version 20.0 | |||||
Sclerocarya birrea leaf and Crinum ornatum extracts showed 75% survival, while Sclerocarya birrea root and Ficus platyphylla extracts exhibited moderate protection (50% survival). The lowest anti-venom activity was observed with Mitragyna inermis root extract, which resulted in 25% survival and reduced mean survival time.
DISCUSSION
Vipers’ venoms are considered poisonous as they contain a large number of toxic components. The venom constitutes major concentrations of zinc-dependent snake venom metalloproteinases (SVMPs), phospholipase-A2 (PLA2s), and serine proteinases, attributing to their hemotoxic and cytotoxic mechanism of action19. Adeyi et al.20 established an estimated lethal dose (LD50) of Echis ocellatus venom to be 0.22 mg/kg−1. Muhammad et al.21 reported an LD50 of (1.24 mg/kg) while, Ernst and Zug22 reported an estimated LD50 of (0.23 mg/kg) in their respective studies. In the present study, the LD50 obtained is 0.316mg/kg b. wt, which is slightly greater than the aforementioned reports. However, it’s lower than the intraperitoneal LD50 reported by Salmanizadeh et al.23 of 11.1 mg/kg. The differences in the LD50 are mainly due to differences in geographical locations of the snakes, sex, diet, and seasonal variation. Additionally, it might be due to differences in the compositions, method of concentration and relative abundance of venom toxins24.
Many reported medicinal plants have antivenom potentials as a result of their phytochemical contents such as flavonoids, phenols, saponnins and alkaloids25. There are reported researches on some plants containing significant levels of bioactive compounds against snakebite like, saponins, alkaloids, tannins, isothiocyanates, flavonoids, phenols as well as vitamins26, these plants include: Calliostoma ornatum, C. nilotica, F. albida, F. platyphylla, S. birrea and M. inermis. Among the snake venom inhibition mechanisms, enzyme inhibition, protein binding as well as free radical inactivation were reported to be possessed by the phytochemicals in those medicinal plants7. The antivenom activity observed in this study might be attributed to the presence of these phytochemicals, possibly exhibiting their effect through the stated mechanisms.
CONCLUSION
This study determined the lethal profile of Echis ocellatus venom, with LD50 and LD100 values established. Several local medicinal plants from Kebbi State, Nigeria, showed significant antivenom activity in experimental models. Some extracts matched or exceeded the efficacy of conventional controls, while others were less effective. These findings validate the traditional use of these plants and provide a foundation for the development of plant-based antivenom therapies. Further studies are recommended to isolate active compounds and explore their mechanisms of action.
SIGNIFICANCE STATEMENT
Snakebite is among the highest neglected health problems in Nigeria that leads to morbidities and mortalities, with rural dwellers being the major victims due to deteriorating modern health facilities. The screened plant extracts used in the present study exhibited substantial efficacy against Echis ocellatus venom. Hence, this research showcases the importance of medicinal plants as an alternative for the development of safe, readily available, and affordable plant-based antivenoms. This may eventually substitute the conventional serum-based antivenins since they possess certain limitations.
ACKNOWLEDGMENT
This research work was fully sponsored by the Nigerian Tertiary Education Trust Fund (TETFund) through Institution-Based Research (IBR) Grant with grant reference number: TETFUND/DESS/UNI/ALIERO/2025/RP/VOL.I.
REFERENCES
- Williams, D.J., M. Abul Faiz, B. Abela-Ridder, S. Ainsworth and T.C. Bulfone et al., 2019. Strategy for a globally coordinated response to a priority neglected tropical disease: Snakebite envenoming. PLoS Negl. Trop. Dis., 13.
- Fernández, C.E.A. and P. Youssef, 2024. Snakebites in the Americas: A neglected problem in public health. Curr. Trop. Med. Rep., 11: 19-27.
- Chippaux, J.P., A. Massougbodji and A.G. Habib, 2019. The WHO strategy for prevention and control of snakebite envenoming: A Sub-Saharan Africa plan. J. Venomous Anim. Toxins Incl. Trop. Dis., 25.
- Sani, U.M., N.M. Jiya, P.K. Ibitoye and M.M. Ahmad, 2013. Presentation and outcome of snake bite among children in Sokoto, North-Western Nigeria. Sahel Med. J., 16: 148-153.
- Nimzing, T.T., Y.A. Arigbede and I. Muhammad, 2022. Examination of snakebite prevalence and factors predispose to bite in Langtang North, Plateau State, Nigeria. FUDMA J. Sci., 6: 130-137.
- Pook, C.E., U. Joger, N. Stümpel and W. Wüster, 2009. When continents collide: Phylogeny, historical biogeography and systematics of the medically important viper genus Echis (Squamata: Serpentes: Viperidae). Mol. Phylogenet. E, 53: 792-807.
- Adeyi, A.O., S.O. Adeyemi, E.O.P. Effiong, B.S. Ajisebiola, O.E. Adeyi and A.S. James, 2021. Moringa oleifera extract extenuates Echis ocellatus venom-induced toxicities, histopathological impairments and inflammation via enhancement of Nrf2 expression in rats. Pathophysiology, 28: 98-115.
- Hamza, M., C. Knudsen, C.A. Gnanathasan, W. Monteiro, M.R. Lewin, A.H. Laustsen and A.G. Habib, 2021. Clinical management of snakebite envenoming: Future perspectives. Toxicon: X, 11.
- Alangode, A., K. Rajan and B.G. Nair, 2020. Snake antivenom: Challenges and alternate approaches. Biochem. Pharmacol., 181.
- Knudsen, C., J.A. Jürgensen, S. Føns, A.M. Haack and R.U.W. Friis et al., 2021. Snakebite envenoming diagnosis and diagnostics. Front. Immunol., 12.
- Gurib-Fakim, A., 2006. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Aspects Med., 27: 1-93.
- Abubakar, M.S., M.I. Sule, U.U. Pateh, E.M. Abdurahman, A.K. Haruna and B.M. Jahun, 2000. In vitro snake venom detoxifying action of the leaf extract of Guiera senegalensis. J. Ethnopharmacol., 69: 253-257.
- Goswami, P.K., M. Samant and R.S. Srivastava, 2014. Snake venom, anti-snake venom & potential of snake venom. Int. J. Pharm. Pharm. Sci., 6: 4-7.
- Razi, M.T., M.H. Hassan Bin Asad, T. Khan, M.Z. Chaudhary, M.T. Ansari, M.A. Arshad and Q. Najam-us Saqib, 2011. Antihaemorrhagic potentials of Fagonia cretica against Naja naja karachiensis (black Pakistan cobra) venom. Nat. Prod. Res., 25: 1902-1907.
- Theakston, R.D.G. and H.A. Reid, 1983. Development of simple standard assay procedures for the characterization of snake venoms. Bull. World Health Organ., 61: 949-956.
- Finney, D.J., 1952. Probit Analysis. 2nd Edn., Cambridge University Press, Cambridge, United Kingdom, Pages: 318.
- Dupont, S., N. Caffin, B. Bhandari and G.A. Dykes, 2006. In vitro antibacterial activity of Australian native herb extracts against food-related bacteria. Food Control, 17: 929-932.
- Premendran, S.J., K.J. Salwe, S. Pathak, R. Brahmane and K. Manimekalai, 2011. Anti-cobra venom activity of plant Andrographis paniculata and its comparison with polyvalent anti-snake venom. J. Nat. Sci. Biol. Med., 2: 198-204.
- Abdullahi, Z.U., S.S. Musa, D. He and U.M. Bello, 2021. Antiprotozoal effect of snake venoms and their fractions: A systematic review. Pathogens, 10.
- Adeyi, A.O., K.K. Mustapha, B.S. Ajisebiola, O.E. Adeyi, D.S. Metibemu and R.E. Okonji, 2022. Inhibition of Echis ocellatus venom metalloprotease by flavonoid-rich ethyl acetate sub-fraction of Moringa oleifera (Lam.) leaves: in vitro and in silico approaches. Toxin Rev., 41: 476-486.
- Muhammad, Y.A., A.A. Bala, S.Y. Magaji, A.I. Doma and A.R. Abubakar et al., 2022. Efficacy testing of commercially available anti-snake venoms against Echis ocellatus venom in Northern Nigeria. Dutse J. Pure Appl. Sci., 8: 116-128.
- Ernst, C.H. and G.R. Zug, 1996. Snakes in Question: The Smithsonian Answer Book. Smithsonian Institution Press, Washington, D.C., USA, ISBN-13: 978-1560986492, Pages: 203.
- Salmanizadeh, H., M. Babaie and H. Zolfagharian, 2013. In vivo evaluation of homeostatic effects of Echis carinatus snake venom in Iran. J. Venomous Anim. Toxins Incl. Trop. Dis., 19.
- Chippaux, J.P., 1998. Snake-bites: Appraisal of the global situation. Bull. World Health Organ., 76: 515-524.
- Kulatunga, W.M.S.S.K. and L.D.A.M. Arawwawala, 2019. Phytochemical analysis of an anti-venom traditional herbal preparation for snake-bite. South Asian Res. J. Nat. Prod., 2: 166-171.
- Saxena, M., J. Saxena, R. Nema, D. Singh and A. Gupta, 2013. Phytochemistry of medicinal plants. J. Pharmacogn. Phytochem., 1: 168-182.
How to Cite this paper?
APA-7 Style
Sani,
I., Ukwuani-Kwaja,
A.N., Kankara,
A.I., Bello,
F., Zubairu,
A. (2026). Antivenom Potentials of Local Medicinal Plants Against Echis ocellatus Envenomation in Aliero, Kebbi State, Nigeria. Trends in Biological Sciences, 2(2), 136-143. https://doi.org/10.21124/tbs.2026.136.143
ACS Style
Sani,
I.; Ukwuani-Kwaja,
A.N.; Kankara,
A.I.; Bello,
F.; Zubairu,
A. Antivenom Potentials of Local Medicinal Plants Against Echis ocellatus Envenomation in Aliero, Kebbi State, Nigeria. Trends Biol. Sci 2026, 2, 136-143. https://doi.org/10.21124/tbs.2026.136.143
AMA Style
Sani
I, Ukwuani-Kwaja
AN, Kankara
AI, Bello
F, Zubairu
A. Antivenom Potentials of Local Medicinal Plants Against Echis ocellatus Envenomation in Aliero, Kebbi State, Nigeria. Trends in Biological Sciences. 2026; 2(2): 136-143. https://doi.org/10.21124/tbs.2026.136.143
Chicago/Turabian Style
Sani, Ibrahim, Angela Nnenna Ukwuani-Kwaja, Aliyu Idris Kankara, Fatima Bello, and Abdulhamid Zubairu.
2026. "Antivenom Potentials of Local Medicinal Plants Against Echis ocellatus Envenomation in Aliero, Kebbi State, Nigeria" Trends in Biological Sciences 2, no. 2: 136-143. https://doi.org/10.21124/tbs.2026.136.143

This work is licensed under a Creative Commons Attribution 4.0 International License.




