Botanical Profiling of Herbal Teas and their Potential as Functional Beverages
| Received 27 Oct, 2025 |
Accepted 10 Feb, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Bioactive compounds in medicinal plants play a key role in disease prevention, particularly in managing diabetes through α-amylase inhibition. This study evaluated the antidiabetic potential of Indian medicinal herbs by assessing the inhibitory activity of three herbal tea formulations (F1, F2, F3) containing Senna auriculata in combination with tulsi, ginger, and cinnamon, respectively. The primary aim was to compare their α-amylase inhibitory efficiency with acarbose and black tea. Materials and Methods: An in vitro assay was conducted using human salivary α-amylase (0.01%) acting on starch slurry at 30, 60, 90, and 120 min at 37°C. Acarbose (Control-1), black tea (Control-2), and formulations F1, F2, and F3 were tested for their inhibitory effects. The FTIR spectroscopy was used to confirm functional groups and the presence of phenolic compounds, while also screening for toxic components. Experiments were in triplicate; data are mean ±SE, analyzed with SPSS 20, p<0.05. Results: The degree of starch hydrolysis followed the order: Black tea >F1>F2>F3. Formulation F3 (Senna auriculata+Cinnamomum zeylanicum) showed the highest α-amylase inhibition (54.70-80.99% within 2 hrs), indicating the strongest antidiabetic potential. F1 and F2 exhibited inhibition ranges of 42.29-67.12 and 42.55-78.24%, respectively. The FTIR spectra confirmed six characteristic phenolic peaks in F1 and F2, while F3 exhibited additional peaks at 4623 cm and 4337.91 cm, indicating higher polyphenol content. No IR absorption between 2220-2250 cm confirmed the absence of cyanide groups, validating the safety of all infusions. Conclusion: Herbal tea formulation F3 (Senna auriculata+Cinnamomum zeylanicum) demonstrated the strongest α-amylase inhibition and highest bioactive compound content, making it the most promising functional beverage for antidiabetic applications. All teas were confirmed safe for consumption. Further in vivo evaluation is recommended to validate therapeutic potential.
| Copyright © 2026 Yasodha and Preethi. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Due to a sedentary lifestyle and occupations, people are prone to type 2 diabetes with an imbalance in glucose metabolism. Rapid digestion of starch by α-amylase is an important factor that leads to the consumption of starch as the main diet repeatedly1,2. There is a quick increase and accumulation of glucose levels in the post-meal blood. Botanicals in herbal tea consumed every day as a functional beverage will be helpful in reducing the risks of Type 2 Diabetes (T2D). Health care needs and drug
discovery depend on herbal medicines. Consuming medicinal plants in trace quantities will reduce the risk of many dieseases including cancer and type 2 diabetes (T2D), which are prevalent among non-communicable diseases3-5. Health benefits of Indian medicinal herbs are well known and play a vital role in the treatment of T2D. The principle of “Let food be thy medicine, and medicine be thy food” was widely accepted in world literature, and herbal tea relationship dates back to the Indian vedas which listed the goodness and abundance of Indian medicinal plants6-10. Herbal teas brewed from all parts of plants contain phenolic bioactive compounds, and consuming herbal teas every day as a functional beverage will help reduce the risk of T2D.
Hence, with this background, the present study was carried out to investigate the botanicals of selected Indian medicinal herbal infusions profiled by FTIR and their inhibitory role against α-amylase during in vitro starch hydrolysis.
MATERIALS AND METHODS
Study area: The present study was conducted at Madha Engineering College during October, 2023-September, 2024 in the Department of Biotechnology. Medicinal herbs were collected from the herbal garden of Madha Engineering College, processed, and stored for further study.
Preparation of herbal tea extracts: Senna auriculata (Tarwar) - flower bud, Ocimum sanctum (Tulsi) - leaves, Zingiber officinale (Ginger) - rhizome from the herbal garden of Madha Engineering College, and Cinnamomum zeylanicum (Cinnamon) - bark from the local market were collected, washed and sun/air dried. About 10 g of black tea was taken as a control. Herbal teas at the ratio of 1:1 w/w were formulated as follows:
| • | F1: Senna auriculata+Ocimum sanctum | |
| • | F2: Senna auriculata+Zingiber officinale | |
| • | F3: Senna auriculata+Cinnamomum zeylanicum |
A 1:50 w/v of dried herbs in fine powder form soaked in hot water at 80-90°C for 5-10 min and filtered afterwards. The extracts of herbal tea formulations (F1, F2 and F3) were used for the experiments of the present study.
FTIR spectral analysis: The FTIR analysis was conducted for the selected Indian medicinal herbal teas viz., Senna auriculata+Ocimum sanctum, Senna auriculata+Zingiber officinale and Senna auriculata+Cinnamomum zeylanicum extracts (F1, F2 and F3, respectively) Fine powder of oven-dried (50°C) samples of selected medicinal herbs were taken. The FTIR spectral analysis of the herbal infusions and black tea was done as per procedures and the pattern recognition followed the standard methods11. The scanning absorption range was 500 to 5000 cm. The peak values of FTIR were recorded for F1, F2 and F3 and black tea (Control). Each and every analysis was repeated twice to confirm the spectrum and the attenuated total reflectance Fourier-transform infrared (ATR-FTIR) spectroscopy of all formulations recorded the secondary metabolites and types of chemical bonds between the chemical molecules of the selected Indian herbs.
In vitro study of inhibitory effect on α-amylase: The inhibitory effect of acarbose (Control-1), black tea (Control-2) and herbal tea formulations (F1, F2 and F3) on α-amylase of human saliva were investigated in vitro. About 10 g of black tea leaves and herbal powders of F1, F2 and F3 were infused in 150 mL hot water. The black tea and herbal solutions were stirred well on a hot plate for 15 minutes at (25±1°C). Then extracts were filtered and centrifuged for 30 min. The filtered extracts were placed in reagent bottles separately and stored at the temperature of 4°C for further analysis. All extractions were done in triplicate (n = 3). Starch hydrolysis by α-amylase was determined as per methodology11. The starch slurry was
prepared using rice starch (10%) in 0.02 M sodium phosphate buffer at pH 6.9. Black Tea extract (Control-2) and herbal tea formulations (F1, F2 and F3) in the proportion of 1: 5 (v/v) aqueous extraction was added into starch. The α-amylase from human saliva (0.01%) was added into starch slurry. The samples were incubated at 37°C. 1 mL of the starch suspension was withdrawn at various time intervals 30, 60, 90, and 120 min. Control tests were carried out using acarbose - C1 and black tea extract - C2. Absorbance was measured at 540 nm.
Percentage of inhibition of α-amylase was calculated as per the formula given by the standard method14:
| OD | = | Optical density | |
| C1 | = | Control sample (without inhibitor) |
Statistical analysis: All experiments were conducted in triplicate. Statistical analysis was carried out using IBM SPSS version 20.0 software. The data expressed as mean±SE and p-value<0.05 was considered significant.
RESULTS AND DISCUSSION
Botanical profiling of our herbal formulations recorded significant and noteworthy results which are discussed in the following sections.
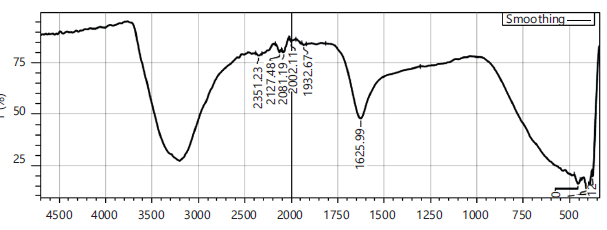
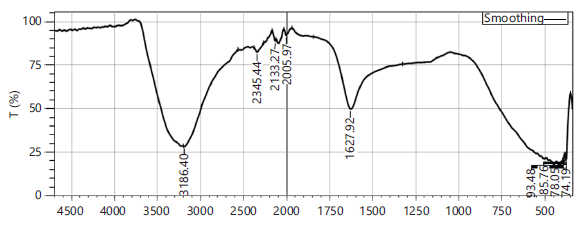
FTIR profiling of herbal teas: The FTIR spectra of black tea (Camellia sinensis) in Control (Fig. 1) is used for comparison. A rapid analysis and confirmation of phytoconstituent structures are useful in the quality checking of herbal teas. The FTIR spectra for black tea (Control) revealed C = C stretching vibration which can be conjugated with C = O exhibited in the region 1626 cm−1 possess only 4 peaks.
For herbal tea formulations F1 and F2, variable stretching vibrations are found in the region between 2002.11 and 2352 cm−1. This illustrates the free amino acids and amino-related functional groups12.
Absorption peaks for Senna auriculata and Ocimum sanctum (F1) at 3186 cm−1 correspond to the -OH stretching vibrations. The broad band at 1627 cm−1 illustrates the confirmation of more phenolic romatic rings compared to the control. The peaks below 100 cm−1 are indicative of low-wavenumber signals that exhibit information about crystal structure, heavy element interactions, and intermolecular forces, providing insights beyond typical functional group analysis.
|
|
|
|
Botanical profiling by FTIR spectra of Senna auriculata and Ocimum sanctum revealed rich phytochemical profiles, with shared peaks indicating common functional groups like hydroxyl (O-H), amine (N-H), carbonyl (C=O), and alkanes, confirming their consortia presence in both Indian herbs. Specific bands for formulation F2 syndicates amides, phenols, alcohols, flavonoids, and carboxylic acids, highlighting its diverse botanicals (Fig. 2), validating the goodness of herbal tea.
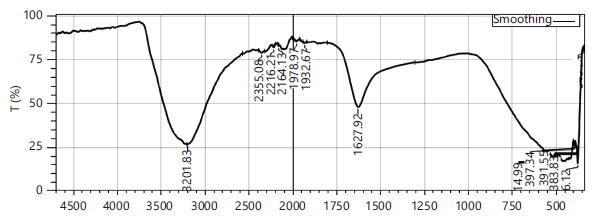
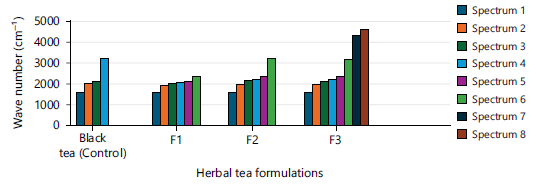
The FTIR spectra of Senna auriculata+Zingiber officinale -F2 (Fig. 3) revealed characteristic peaks indicating botanical profile with noteworthy stretching bands for O-H (alcohols/phenols) around 3300-3400 cm−1, C-H stretching (alkanes/alkenes) near 2900 cm−1, C=C stretching (aromatics/alkenes) around 1600-1650 cm−1, and C-O stretching (ethers/alcohols/phenols) in the 1000-1200 cm−1 range, confirming presence of compounds like gingerols, shogaols, polysaccharides, and flavonoids. The FTIR spectra of F1, F2 and F3 are illustrated in Fig. 1-4.
|
| Table 1: | Alpha amylase inhibition activity (%) of acarbose, black tea and herbal teas | |||
| Time (minutes) | C1 | C2 | F1 | F2 | F3 |
| 30 | 55.02 | 33.5 | 42.29 | 42.55 | 54.7 |
| 60 | 62.75 | 45.21 | 52.66 | 53 | 65.15 |
| 90 | 77.33 | 52.99 | 59.08 | 64.05 | 74.01 |
| 120 | 86.11 | 60.3 | 67.12 | 78.24 | 80.99 |
| S.E | 1.3 | 1.21 | 1.17 | 1.6 | 1.1 |
| C.D | 0.02 | 0.04 | 0.02 | 0.03 | 0.03 |
| Mean values of triplicates and p-value is significant at <0.05, S.E: Standard error, C.D: Critical difference at p<0.05, C1: Acarbose, C2: Black tea, F1: Senna auriculata+Ocimum sanctum, F2: Senna auriculata+Zingiber officinale and F3: Senna auriculata+Cinnamomum zeylanicum | |||||
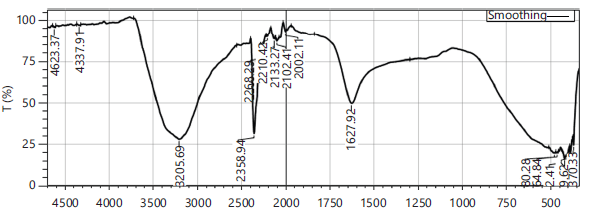
Botanical profiling of Senna auriculata and Cinnamomum zeylanicum possess a peak at 1627.92 cm−1 of spectral wave number showed the stretching vibration of C=O (carbonyl) bonds from flavonoids, polyphenols, and catechins. Uptake in the area of 1932.67-1979 cm−1 showed stretching of aromatic compounds. Uptake in the area of 2002-2210 cm−1 from the Fig. 4 indicated the vibration of the C=C bond of aromatic compounds. Maximum intensity peaks in the area of 2133-2164 cm−1 revealed their silicon compounds and carboxyl acid confirmed their suitability in functional beverages. Significant absorbance differences at specific peaks between the range 4388 and 4623 cm−1 indicates potential for detecting variations related to O-H bending and water content. The FTIR peaks below 750 cm−1 signify the stretching of aromatic ring vibrations (Fig. 4).
In F1 and F2 formulations 6 peaks were found (Fig. 5). Absorption bands correspond to 8 peaks referred to the functional groups present in F3 infusion are existing between 4623 to 4337 cm-112-14. Thus the formulation (F3) of Senna auriculata and Cinnamomum zeylanicum had depicted four similar peaks to that of control resembling the phytophenols and other botanicals. Furthermore F3 is analogous to F1 and F2 with two peaks between 2355 to 3208 cm-1 illustrated in Fig. 5. Furthermore there are two new peaks in F3 recorded their unique phytochemicals. The presence of botanicals in Senna auriculata and Cinnamomum zeylanicum confirmed by the strong stretching of 8 peaks for their vital functional groups with medicinal values present in F3 infusion (Fig. 5).
Inhibitory effect on α-amylase: In the present study, aqua extracts of F1 F2 and F3 were assessed for inhibition of α-amylase effects during in vitro starch break down. Standard drug acarbose (Control-1) and black tea (Control-2) were used for comparison. Acarbose is a synthetic inhibitor of starch digestive enzyme α-amylase used as a standard drug in the assessment of α-amylase inhibitory study15-19. In our present study the herbal botanicals with reference to phenolics in F1, F2 and F3 showed an effective inhibition against α-amylase (Table 1). The results were compared with acarbose (C1).
Formulation of Senna auriculata in combination with tulsi (F1) and ginger (F2) inhibit the activity of α-amylase at the rate of 42.29-67.12% and 42.55-78.24%, respectively (Table 1). The conformity on structures of phenolic compounds in F1 and F2 was revealed by six peaks in FTIR spectra. A significantly comparable results pertaining to (s) low starch hydrolysis and high percentage of inhibitory effect on α-amylase were obtained in the herbal tea formulations F1, F2 and F3 with contributions ranging from 42.29% (F1) to 80.99% (F3). The inhibitory effect of black tea and F1 was reached in 120 minutes upto 60.30 and 67.12%, respectively. Whereas F3- Senna auriculata+Cinnamomum zeylanicum was efficiently inhibit the α-amylase ranging from 54.70 to 80.99% in 2 hrs time. Earlier researchers20-23 experimented the inhibitory activity of alpha amylase and alpha glucosidase enzyme by various botanicals supports the performance of selected Indian herbs in the present study.
An appreciable role of F3- Senna auriculata+Cinnamomum zeylanicum with more functional groups(polyphenols) was confirmed through FTIR. This infusion effected a superior potentiality contributed to the antidiabetic activity through α-amylase inhibition. The FTIR characterization of functional groups of several medicinal plants was done by earlier researchers supports the present investigation24-26.
Experiments pertaining to the in vitro inhibitory effect on α-amylase during hydrolysis by different plant sourced bioactive compounds were studied globally. Among the botanicals, phenolic compounds are very important and have greater potentiality to act against α-amylase than the synthetic drugs. The negative relationship between the heterogenous polyphenols and in vitro rate of starch digestion is due to the number and position of hydroxyl groups of the phenolic compounds. This could prevent starch gelatinization in turn it inhibits α-amylase activity27,28.
CONCLUSION
Antidiabetic activity through α-amylase inhibition by all herbal teas in F1, F2 and F3 was significantly comparable with the synthetic drug acarbose (55-86%). However the superior inhibitory effect of F3 (Senna auriculata+Cinnamomum zeylanicum) on α-amylase was ranging from 54.70 to 80.99% in 2 hrs time. The in vitro investigation of α-amylase activity during starch hydrolysis revealed the slow release of starch and high rate of inhibition by all herbal teas at the rate of 42.29 to 80.99% in 2 hrs time. It can be concluded that scientific evidence of the antidiabetic effect of herbal teas lead towards the recommendation of Senna auriculata (Tarwar) in fusion with tulsi, ginger and cinnamon as herbal remedies for T2D treatment. Hence the phenolic compounds in all herbal tea formulations proved its functional role in controlling the α-amylase during in vitro starch hydrolysis. Further research would be directed towards experimenting the potentiality of herbal teas in post meal glycaemic control of type 2 diabetics in clinical research.
SIGNIFICANCE STATEMENT
This study highlights the potential of Indian medicinal herbs as functional antidiabetic beverages. Among the tested formulations, F3 (Senna auriculata+Cinnamomum zeylanicum) exhibited the strongest α amylase inhibition and highest polyphenol content, indicating superior bioactivity. The findings validate the efficacy and safety of herbal teas in managing postprandial hyperglycemia, offering a natural, accessible alternative to synthetic inhibitors like acarbose. These results provide a scientific basis for developing functional drinks with therapeutic potential against diabetes.
ACKNOWLEDGMENT
The authors are grateful to Tamil Nadu State Council of Science and Technology, Chennai for the financial assistance to conduct this research.
REFERENCES
- Olokoba, A.B., O.A. Obateru and L.B. Olokoba, 2012. Type 2 diabetes mellitus: A review of current trends. Oman Med. J., 27: 269-273.
- Khan, S., M. Markoulli, A.T. Tsoi and M. Willcox, 2025. Exploring the efficacy of plant-based nutraceuticals in managing diabetic neuropathy. Inflammopharmacology, 33: 2935-2949.
- McKay, D.L. and J.B. Blumberg, 2002. The role of tea in human health: An update. J. Am. Coll. Nutr., 21: 1-13.
- Wargovich, M.J., C. Woods, D.M. Hollis and M.E. Zander, 2001. Herbals, cancer prevention and health. J. Nutr., 131: 3034S-3036S.
- Cabrera, C., R. Artacho and R. Giménez, 2006. Beneficial effects of green tea-a review. J. Am. Coll. Nutr., 25: 79-99.
- Jain, S.R. and S.N. Sharma, 1967. Hypoglycaemic drugs of Indian indigenous origin. Planta Med., 15: 439-442.
- Samy, R.P. and S. Ignacimuthu, 2000. Antibacterial activity of some folklore medicinal plants used by tribals in Western Ghats of India. J. Ethnopharmacol., 69: 63-71.
- Akhtar, S., F. Hassan, S.R. Saqlain, A. Ali and S. Hussain, 2023. The prevalence of peripheral neuropathy among the patients with diabetes in Pakistan: A systematic review and meta-analysis. Sci. Rep., 13.
- Ahmed, M.F., H. Thayyil, A.S. Rasheed and M. Ibrahim, 2010. Anti-ulcer activity of Cassia auriculata leaf extract. Pharmacogn. J., 2: 53-57.
- Singh, D., P. Singh, A. Gupta, S. Solanki, E. Sharma and R. Nema, 2012. Qualitative estimation of bioactive compound present in Centella asiatica: An important medicinal plant. Int. J. Life Sci. Med. Res., 2: 5-7.
- Aboulwafa, M.M., F.S. Youssef, H.A. Gad, S.D. Sarker, L. Nahar, M.M. Al-Azizi and M.L. Ashour, 2019. Authentication and discrimination of green tea samples using UV-vis, FTIR and HPLC techniques coupled with chemometrics analysis. J. Pharm. Biomed. Anal., 164: 653-658.
- Senthilkumar, S.R., T. Sivakumar, K.T. Arulmozhi and N. Mythili, 2017. FT-IR analysis and correlation studies on the antioxidant activity, total phenolics and total flavonoids of Indian commercial teas (Camellia sinensis L.)-a novel approach. Int. Res. J. Biol. Sci., 6: 1-7.
- Koh, L.W., L.L. Wong, Y.Y. Loo, S. Kasapis and D. Huang, 2010. Evaluation of different teas against starch digestibility by mammalian glycosidases. J. Agric. Food Chem., 58: 148-154.
- Xiao, J., G. Kai, X. Ni, F. Yang and X. Chen, 2011. Interaction of natural polyphenols with α-amylase in vitro: Molecular property-affinity relationship aspect. Mol. BioSyst., 7: 1883-1890.
- Liu, Y., C. Guo, E. Zang, R. Shi and Q. Liu et al., 2023. Review on herbal tea as a functional food: Classification, active compounds, biological activity, and industrial status. J. Future Foods, 3: 206-219.
- Intrasook, J., T.W. Tsusaka and A.K. Anal, 2024. Trends and current food safety regulations and policies for functional foods and beverages containing botanicals. J. Food Drug Anal., 32: 118-145.
- Matsui, T., T. Tanaka, S. Tamura, A. Toshima and K. Tamaya et al., 2007. α-glucosidase inhibitory profile of catechins and theaflavins. J. Agric. Food Chem., 55: 99-105.
- Forester, S.C., Y. Gu and J.D. Lambert, 2012. Inhibition of starch digestion by the green tea polyphenol, (-)‐epigallocatechin‐3‐gallate. Mol. Nutr. Food Res., 56: 1647-1654.
- Kan, L., E. Capuano, V. Fogliano, T. Oliviero and R. Verkerk, 2020. Tea polyphenols as a strategy to control starch digestion in bread: The effects of polyphenol type and gluten. Food Funct., 11: 5933-5943.
- Kozłowicz, K., R. Różyło, B. Gładyszewska, A. Matwijczuk and G. Gładyszewski et al., 2020. Identification of sugars and phenolic compounds in honey powders with the use of GC-MS, FTIR spectroscopy, and X-ray diffraction. Sci. Rep., 10.
- Vanapatla, S.R. and F. Begam, 2019. Evaluation of α-amylase and α-glucosidase enzyme inhibitory activities of trichuriella monsoniae bennet. Int. J. Pharm. Sci. Res., 10: 1342-1346.
- LIczbiński, P. and B. Bukowska, 2022. Tea and coffee polyphenols and their biological properties based on the latest in vitro investigations. Ind. Crops Prod., 175.
- Kwon, Y.I., E. Apostolidis and K. Shetty, 2008. Inhibitory potential of wine and tea against α‐Amylase and α‐Glucosidase for management of hyperglycemia linked to type 2 diabetes. J. Food Biochem., 32: 15-31.
- Gorgulu, S.T., M. Dogan and F. Severcan, 2007. The characterization and differentiation of higher plants by Fourier transform infrared spectroscopy. Appl. Spectrosc., 61: 300-308.
- Kumar, J.K. and A.G.D. Prasad, 2011. Identification and comparison of biomolecules in medicinal plants of Tephrosia tinctoria and Atylosia albicans by using FTIR. Rom. J. Biophys., 21: 63-71.
- Saxena, M. and J. Saxena, 2012. Evalution of phytoconstituents of Acorus calamus by FTIR and UV-VIS spectroscopic analysis. Int. J. Biol. Pharm. Res., 3: 498-501.
- Veeramani, S., A.P. Narayanan, K. Yuvaraj, R. Sivaramakrishnan and A. Pugazhendhi et al., 2022. Nigella sativa flavonoids surface coated gold NPs (Au-NPs) enhancing antioxidant and anti-diabetic activity. Process Biochem., 114: 193-202.
- Saidu, S., C.O. Eleazu, D. Ebuka, A. Ikechukwu, M. Blessing, N. Chibuike and C. Chukwuma, 2017. Starch hydrolysis, polyphenol contents and in vitro alpha amylase inhibitory properties of some Nigerian foods as affected by cooking. Front. Nutr., 4.
How to Cite this paper?
APA-7 Style
Yasodha,
T., Preethi,
D. (2026). Botanical Profiling of Herbal Teas and their Potential as Functional Beverages. Trends in Biological Sciences, 2(1), 117-124. https://doi.org/10.21124/tbs.2026.117.124
ACS Style
Yasodha,
T.; Preethi,
D. Botanical Profiling of Herbal Teas and their Potential as Functional Beverages. Trends Biol. Sci 2026, 2, 117-124. https://doi.org/10.21124/tbs.2026.117.124
AMA Style
Yasodha
T, Preethi
D. Botanical Profiling of Herbal Teas and their Potential as Functional Beverages. Trends in Biological Sciences. 2026; 2(1): 117-124. https://doi.org/10.21124/tbs.2026.117.124
Chicago/Turabian Style
Yasodha, T., and D. Preethi.
2026. "Botanical Profiling of Herbal Teas and their Potential as Functional Beverages" Trends in Biological Sciences 2, no. 1: 117-124. https://doi.org/10.21124/tbs.2026.117.124

This work is licensed under a Creative Commons Attribution 4.0 International License.




