Isolation and Identification of Entomopathogenic Fungi from Chili Rhizosphere Against Fall Armyworm
| Received 26 Oct, 2025 |
Accepted 10 Apr, 2026 |
Published 30 Jun, 2026 |
Background and Objective: Chili (Capsicum annuum L.) is an economically important horticultural crop that is frequently damaged by Spodoptera larvae, leading to substantial yield losses. Pest management in chili cultivation relies heavily on chemical pesticides, which pose environmental and health risks. Although entomopathogenic fungi are known to be diverse in Indonesian soils, their potential as biological control agents in chili rhizospheres remains underexplored. This study aimed to isolate and identify entomopathogenic fungi from the chili rhizosphere and to evaluate their pathogenicity against Spodoptera larvae. Materials and Methods: Soil samples were collected from the rhizosphere of chili plants and cultured to isolate fungal species. Fungal isolates were identified based on macroscopic and microscopic morphological characteristics using standard taxonomic keys. Pathogenicity assays were conducted following Koch’s postulates by inoculating Spodoptera larvae with fungal suspensions at a concentration of 107 conidia/mL. Larval mortality was observed daily to assess pathogenic effects. Data were analyzed descriptively and quantitatively, with larval mortality expressed as mean percentage and evaluated using one-way ANOVA followed by Duncan’s Multiple Range Test at α = 0.05. Results: Three fungal genera, Trichoderma sp., Aspergillus sp., and Penicillium sp. were successfully isolated from the chili rhizosphere. Morphological features of the isolates were consistent with previously reported descriptions in the literature. All isolates caused mortality in Spodoptera larvae, with average death occurring on the fourth day after inoculation. Mortality was mainly associated with fungal secondary metabolites rather than visible external mycelial growth, suggesting opportunistic entomopathogenic behavior. Conclusion:The findings demonstrate that fungi inhabiting the chili rhizosphere possess pathogenic activity against Spodoptera larvae and represent promising candidates for biological control. These results support the potential use of indigenous soil fungi as environmentally friendly alternatives to chemical pesticides. Further studies focusing on metabolite characterization, field efficacy, and non-target effects are recommended to advance their application in sustainable pest management.
| Copyright © 2026 Mutia et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Indonesia is known for its high biodiversity, including a rich diversity of entomopathogenic fungi. This diversity is supported by the country’s tropical geography, characterized by a relatively stable temperature throughout the year, two distinct seasons, and various habitat types. Numerous studies on entomopathogenic fungi have been conducted worldwide. In the tropical rainforests of Thailand, for example, 15 genera of entomopathogenic fungi have been recorded, of which 13 genera have been successfully isolated. In Indonesia, nine genera have been reported from Telaga Warna and three from Cangkuang1.
Red chili (Capsicum annuum L.) is one of the most economically valuable horticultural commodities and plays a strategic role in Indonesian agriculture. Its cultivation remains highly prospective, yet it is also one of the commodities that frequently contributes to national inflation due to price fluctuations. With the growing population and the expansion of chili-based food industries, the demand for chili continues to increase annually. Consequently, chili cultivation offers substantial economic potential but also requires considerable capital investment and technical skills2.
Most chili farmers in Indonesia have not yet implemented Integrated Pest Management (IPM) techniques due to limited knowledge and technical capacity. Consequently, excessive use of chemical pesticides remains a common practice to prevent yield loss or crop failure. However, this overreliance on synthetic pesticides poses serious environmental risks. Ideally, pesticide application should follow the six principles of proper use target, quality, type, timing, dosage, and method2.
According to the concept of IPM, pest control can be achieved through various approaches, including cultural, physical, mechanical, biological, and chemical methods3. Biological control agents encompass a wide range of organisms such as parasitoids, viruses, entomopathogenic fungi, plant-derived biopesticides, natural enemies, and refugia plants4.
One promising approach is the utilization of biological agents such as entomopathogenic fungi. These microorganisms are capable of infecting and killing insect pests naturally and can be found in diverse ecosystems, including the rhizosphere the soil zone surrounding plant roots. The rhizosphere is a rich and dynamic habitat for microorganisms that not only promotes plant growth but also contributes to plant defense against pests and pathogens.
The presence of entomopathogenic fungi in the rhizosphere of chili plants functions not only as a biocontrol mechanism but also enhances soil health and supports plant growth. Previous studies have shown that these fungi contribute to natural pest suppression and improved crop yield5. Furthermore, the exploration of entomopathogenic fungi in various ecosystems, including agricultural lands, has demonstrated their great potential for developing effective and sustainable biopesticides6.
Isolation of entomopathogenic fungi can be performed through several methods, one of which involves using insect larvae as bait. Thalib et al.7 reported that entomopathogenic fungi isolated from agricultural soils were effective against pests such as Scirpophaga incertulas. Similarly, several entomopathogenic fungi from farmlands that can be used to control pests in both food and horticultural crops. These isolation methods not only enable the discovery of active fungal strains but also provide insights into the species diversity present in the plant rhizosphere8.
The isolation and identification of entomopathogenic fungi from the rhizosphere of chili plants represent an important step toward exploring their potential as biological control agents. By identifying the fungal species inhabiting the rhizosphere, it becomes possible to develop more effective, safe, and sustainable pest management strategies. Such efforts not only support agricultural sustainability but also help maintain chili crop productivity to meet the growing demands of Indonesian society. This study aimed to isolate and identify entomopathogenic fungi from the rhizosphere of chili (Capsicum annuum L.) plants and to evaluate their pathogenicity against Spodoptera larvae as potential biological control agents.
MATERIALS AND METHODS
Type of research: This study was exploratory and descriptive, aiming to isolate and identify entomopathogenic fungi from the rhizosphere of chili plants (Capsicum annuum L.) and to assess their potential as biological control agents against Spodoptera sp. larvae. Soil sampling was conducted at the UPT of Food and Horticultural Crop Protection, Maros Regency, while fungal isolation, Koch’s postulate testing, and identification were performed at the Biology Laboratory, Faculty of Mathematics and Natural Sciences, Universitas Negeri Makassar. The study was conducted from February to August 2025.
Tools and materials: The main equipment included a small shovel, plastic bags, Petri dishes, test tubes, a vortex mixer, a hot plate, an autoclave, a microscope, and a laminar air flow cabinet. Materials used were Potato Dextrose Agar (PDA), chloramphenicol, sterile distilled water, 70% alcohol, sterile cotton, aluminum foil, filter paper, baby corn, and third-instar Spodoptera sp. larvae.
Soil sampling: Soil samples were collected from the rhizosphere of chili plants at five diagonal points in the field. Samples were taken 10-15 cm deep and approximately 100 g from each point, then pooled into a composite sample of about 1 kg. The samples were labeled according to location and date, stored in sterile plastic bags, and transported to the laboratory for fungal isolation9.
Isolation of entomopathogenic fungi: Ten grams of composite soil were suspended in 100 mL of sterile distilled water and serially diluted up to 10–7. From dilutions 10–3 to 10–7, 1 mL aliquots were spread onto PDA plates using the spread plate technique and incubated at room temperature. Fungal colonies with different morphological characteristics were selected and purified by subculturing on fresh PDA media.
Purification and culturing: Distinct colonies were transferred to new PDA plates for purification. The PDA media were prepared by dissolving 19 g of PDA powder in 500 mL of distilled water, supplemented with 1 g chloramphenicol, and sterilized at 121°C for 15 min under 1 atm pressure. Plates were poured under aseptic conditions and incubated for four days before further analysis.
Rearing of test insects: Third-instar larvae of Spodoptera sp. were collected from corn fields and reared in the laboratory. Larvae were maintained in aerated plastic containers and fed pesticide-free baby corn daily. Upon pupation, pupae were transferred to containers filled with sterilized sand until adult emergence. Adults were allowed to mate and oviposit on wax paper, and the resulting eggs were reared until third-instar larvae for pathogenicity testing10.
Pathogenicity test (Koch’s Postulate): Pathogenicity was tested using a larval dipping method10. Spore suspensions (107 conidia/mL) were prepared by flooding seven-day-old fungal cultures with sterile distilled water and homogenizing with a vortex mixer. Spore density was determined using a haemocytometer following Oktariana et al11:
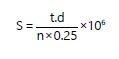 |
Description:
| S | = | Spore count/mL | |
| t | = | Total spores observed | |
| n | = | Number of counting chambers | |
| 0.25 | = | Correction factor |
Each isolate was tested on five third-instar Spodoptera larvae, which were dipped briefly in the suspension and transferred to sterile containers containing fresh baby corn. Mortality was recorded daily to confirm pathogenicity.
Identification of fungi: Fungal identification was conducted using macroscopic and microscopic observations. Slide cultures were prepared12 to observe conidiophore branching, spore shape, and hyphal structure under a compound microscope (40×magnification). Colony morphology, including color, texture, and growth pattern, was recorded after seven days of incubation. Identification was made to the genus level using morphological keys by Barnett and Hunter13.
Data analysis: Data were analyzed descriptively and quantitatively. Macroscopic and microscopic characteristics of fungal isolates were described to determine their morphological features and genus-level identification. Larval mortality data obtained from the pathogenicity test were expressed as percentage mortality and presented as mean values. Differences in larval mortality among fungal isolates were analyzed using one-way analysis of variance (ANOVA). When significant differences were detected, comparisons among treatments were conducted using Duncan’s Multiple Range Test (DMRT). Statistical analyses were performed at a significance level of α = 0.05.
RESULTS AND DISCUSSION
Identification of entomopathogenic fungi in the rhizosphere of chili plants: Isolates of entomopathogenic fungi from rhizosphere soil were obtained using the spread plate method on Potato Dextrose Agar (PDA) medium. Four isolates were successfully recovered from the rhizosphere of chili plants. The identification of entomopathogenic fungi represents a crucial preliminary step to determine the diversity of fungal species inhabiting the chili rhizosphere. The rhizosphere was chosen because this region is known to be rich in microbial communities, including soil fungi with potential entomopathogenic properties. Through isolation and morphological observation both macroscopic and microscopic the characteristics and diversity of the isolated fungi were determined. This identification forms the basis for assessing the pathogenic potential of each isolate against Spodoptera larvae in subsequent bioassays. The morphological characteristics of the entomopathogenic fungal isolates obtained from the chili rhizosphere are summarized in Table 1. Colony images were used to document the overall growth pattern of each isolate on PDA medium. Microscopic features, including hyphal structure and conidiophore morphology, were observed to support genus-level identification. Colony color, shape, and margin provided important diagnostic traits commonly used in fungal taxonomy. Spore characteristics, such as shape and arrangement, served as key indicators for distinguishing fungal genera, while colony texture reflected differences in mycelial density and sporulation capacity among the isolates.
Isolate J1-Trichoderma sp.: Based on the results, isolate J1 was identified as a fungus belonging to the genus Trichoderma sp. Macroscopically, the colony initially appeared white; by day 5, the mycelium turned green with a slight white margin, and by day 7, the colony became entirely green with a green reverse side. The colony was circular with regular margins and evenly spread. Its surface texture was coarse and powdery, and the colony was relatively thin.
Microscopically (40×10 magnification), J1 exhibited branched, septate hyphae and conidia that were globose and clustered at the tips of the conidiophores. Unlike the elongated, septate spores typical of pathogenic genera such as Fusarium, Trichoderma conidia are round, green, and form chains a key diagnostic feature. The conidia are asexual (unicellular), usually globose to subglobose, occasionally elliptical, depending on species. They develop on branched, verticillate conidiophores that form a characteristic “Christmas-tree” structure under the microscope. The conidial wall is thin and smooth when immature (hyaline), thickening and turning light to dark green upon maturity due to pigment accumulation such as viridin, giving the colonies their typical green coloration on PDA or malt extract agar14.
| Table 1: | Results of morphological characterization of entomopathogenic fungi in the rhizosphere of chili plants | |||
| Isolate | Colony image | Microscopic | Description | Colony color | Colony shape and margin | Spore | Colony texture |
| J1 | 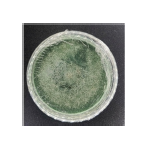 |
 |
Hyphae Conidiophore Conidia |
Dark green | Circular and evenly spread |
Conidiospore | Powdery |
| J2 | 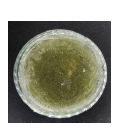 |
 |
Conidia Conidiophore |
Yellowish green | Circular and flat | Conidiospore | Powdery |
| J3 | 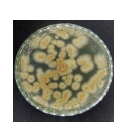 |
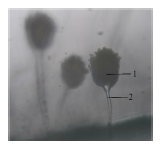 |
Conidia Conidiophore |
Brownish yellow |
Conidiospore | Powdery | |
| J4 | 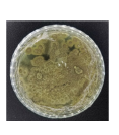 |
 |
Conidia Conidiophore Hyphae |
Dartmouth green (Dark green) |
Concentric circular | Conidiospore | Velvety |
According to Samson et al.15, Trichoderma species show rapid colony growth, initially white and later turning green. Their conidiophores are branched with cylindrical phialides, and conidia are globose to subglobose, hyaline, and either smooth or rough. Similarly, Watanabe16 described Trichoderma colonies as initially white, then green to yellowish-green in mature mycelial phases. Conidiophores are irregularly branched, forming a pyramidal structure, and the conidia are transparent, hyaline, smooth, and round to oval in shape.
Trichoderma (Ascomycota, Hypocreales) reproduces primarily asexually, with conidia as its main spore type; sexual forms are rarely observed. Although, Trichoderma is widely recognized as a plant-pathogen antagonist, certain strains can produce hydrolytic enzymes (chitinases, proteases, cellulases) and secondary metabolites toxic to insects. This explains the observed larval mortality of Spodoptera sp. in this study. Harman et al.17 reported that peptaibols produced by Trichoderma can disrupt cell membranes, including insect tissues. Thus, the presence of this isolate in chili rhizospheres indicates dual potential as a plant disease biocontrol agent and as an opportunistic entomopathogen.
Isolate J2 was identified as a fungus belonging to the genus Aspergillus sp. Macroscopically, the colonies appeared yellowish-green with white margins, turning completely yellowish-green by day 7. The surface texture was powdery, with rapid and uneven radial growth.
Microscopic observation revealed septate hyphae and long, slender conidiophores that terminated in a swollen vesicle. Conidia were globose, forming chains at the tip of phialides. The main spores are asexual, unicellular conidia globose to subglobose with roughened walls (echinulate or reticulate in some species). They form catenately at the phialide tips arising from vesicles at the end of unbranched conidiophores, producing the characteristic “aspergillum” structure18.
Watanabe16 described Aspergillus conidiophores as hyaline, erect, thick-walled, and simple, producing black masses of conidia. On agar media, colonies typically appear uniformly black. According to Nyongesa et al.19, Aspergillus colonies are dark green with a flat, rough surface. Conidiophores are smooth, hyaline, and unbranched, ending in swollen vesicles that bear phialides producing globose conidia.
Aspergillus mainly reproduces asexually via abundant conidia, while sexual reproduction is rarely observed under laboratory conditions. Though Aspergillus is not an obligate entomopathogen, certain species produce toxic secondary metabolites such as aflatoxin, gliotoxin, and organic acids that disrupt insect physiology and lead to accelerated larval mortality. These findings support the notion that Aspergillus can act as an opportunistic pathogen, particularly under humid environmental conditions that promote spore germination and mycelial growth. Thus, the Aspergillus sp. isolate obtained from chili rhizospheres may contribute to Spodoptera larval mortality through metabolic toxicity rather than direct fungal infection. Such as Beauveria bassiana isolated from plant roots and combined with botanical pesticides can increase mortality Spodoptera frugiperda20.
Isolate J3-Aspergillus sp.: Isolate J3 was also identified as belonging to the genus Aspergillus sp. Macroscopically, the colony center appeared brown with white margins, later becoming yellowish-brown with persistent white edges by day 7. The colony surface was velvety, thin, and spread evenly outward.
Microscopically, J3 exhibited septate, hyaline hyphae and single conidiophores bearing round, black conidia. According to Watanabe16, Aspergillus conidiophores are pale brown, septate, and unbranched. Fadhilah et al.21 described Aspergillus sp. as a eukaryotic microorganism within Ascomycetes, characterized by branched, septate hyphae, conidiophores arising from swollen foot cells, and sterigmata producing chains of green, brown, or black conidia.
Macroscopically, Aspergillus colonies are fibrous, smooth, and convex with color variations from gray-green to brownish-green, black, or white. Colony color often correlates with spore pigmentation. Morphological variations among the Aspergillus isolates observed in this study likely reflect species-level differences and environmental growth conditions.
Regarding pathogenicity, similar to isolate J2, Aspergillus sp. in J3 is not a primary entomopathogen but can still induce Spodoptera larval mortality via toxic metabolites. The combination of opportunistic infection through hyphal penetration and toxin production accelerates larval death. Thus, Aspergillus isolates from chili rhizospheres may function as opportunistic insect pathogens.
Isolate J4-Penicillium sp.: Isolate J4 was identified as belonging to the genus Penicillium sp. Macroscopically, the colonies appeared green with white margins initially, becoming dark green with a slight white edge by day 7. The surface was velvety, and the colonies were moderately thick.
Microscopically, the fungus exhibited branched, septate, hyaline hyphae. Conidiophores were unseptate, hyaline, and branched, forming brush-like (penicillus) structures with two metulae bearing phialides, each producing globose conidia in chains. The conidia are asexual, unicellular, and globose to subglobose, with smooth or finely roughened walls13,22.
According to Anggraeni and Usman22, Penicillium colonies are initially white and later turn bluish-green, gray-green, or olive-gray, occasionally yellowish or reddish, with pale yellow reverses. Microscopically, the fungus has hyaline hyphae, globose conidia, and clustered phialides. Barnett and Hunter13 noted that conidiophores can arise singly or as synnemata, with penicillate branching forming groups of flask-shaped phialides.
Penicillium (Ascomycota, Eurotiales) is a ubiquitous filamentous fungus found in soil, food, and air, widely known as the source of the antibiotic penicillin (P. notatum), as well as an organic decomposer and food spoilage organism. Its main spores are conidia with a characteristic brush-like “penicillus” morphology, similar to Aspergillus but more complex23.
Although Penicillium is generally saprophytic, several species can produce toxic secondary metabolites such as patulin, citrinin, and citreoviridin that are harmful to insects and other organisms. This may explain the larval mortality observed in Spodoptera following inoculation with this isolate. Hence, Penicillium sp. from chili rhizospheres may act as an opportunistic entomopathogen, causing insect mortality through metabolite toxicity and possible secondary infection.
Koch’s postulate test: To confirm that Spodoptera larval mortality was caused by the isolated fungi, Koch’s postulate test was conducted. Four fungal isolates were re-inoculated into healthy larvae. Reisolation from infected larvae grown on PDA yielded colonies identical to the inoculated fungi, confirming causality. The results of the Koch’s postulate test are presented in Table 2. Re-isolation of fungi from infected Spodoptera larvae showed that the morphological characteristics of the re-isolated colonies were consistent with those of the original isolates. This result confirms that larval mortality was caused by the applied entomopathogenic fungal isolates.
Koch’s postulate remains the fundamental principle for determining causal relationships between a microorganism and the disease it induces. In this study, fungal suspensions at a concentration of 107 conidia/mL were used, following the standard bioassay range for entomopathogenic fungi (106-108 conidia/mL)24,25. This concentration ensures sufficient inoculum for infection or toxin exposure while preventing false negatives due to inadequate spore contact26-29.
| Table 2: | Results of reisolation of Spodoptera sp. larvae after application of fungus | |||
| Isolate name | Infected larvae | Reisolation results of Spodoptera sp. larvae |
| J1 |  |
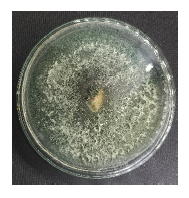
|
| J2 |  |
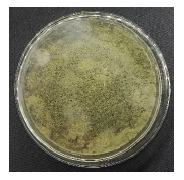 |
| J3 |  |
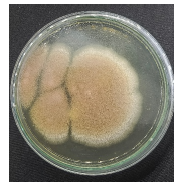 |
| J4 |  |
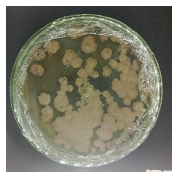 |
| Isolate J1: Trichoderma sp., Isolate J2: Aspergillus sp., Isolate J3: Aspergillus sp. and Isolate J4: Penicillium sp. | ||
The dipping method was employed for inoculation, as it allows uniform spore contact over the entire larval surface, enhancing infection likelihood and ensuring reproducibility. This approach has been shown to yield more consistent infection rates than spraying, especially in laboratory-scale studies24.
Larval death typically occurred four days after inoculation, characterized by body darkening, hardening, shrinkage, and desiccation, though no mycelial growth was visible on the cadavers. This may result from (1) toxic secondary metabolites causing systemic poisoning, (2) absence of specialized infective structures (e.g., appressoria), unlike obligate entomopathogens such as Beauveria bassiana and Metarhizium anisopliae that produce dense external mycelia24,25, or (3) suboptimal humidity preventing external mycelial development despite internal infection.
Hence, the observed larval mortality likely resulted from the toxicity of secondary metabolites or internal infection, rather than external mycelial growth. This finding indicates that chili rhizosphere fungi act as opportunistic entomopathogens, differing from obligate species that exhibit external mycelial formation post-host death.
Opportunistic entomopathogens typically cause insect mortality through sequential stages: (1) Spore contact with larval surfaces, (2) Production of toxic metabolites (e.g., aflatoxin, gliotoxin, patulin), (3) Entry through natural openings (spiracles, mouth, wounds), and (4) Physiological disruption leading to tissue degradation and death. Unlike true entomopathogens that produce external mycelium, opportunistic fungi induce death primarily via metabolite toxicity rather than direct mycelial invasion.
CONCLUSION
This study isolated and identified three fungal genera, Trichoderma sp., Aspergillus sp., and Penicillium sp., from the chili plant rhizosphere (Capsicum annuum L.). Pathogenicity tests showed that all isolates caused larval mortality of Spodoptera sp. by the fourth day after inoculation at 107 conidia/mL. The mortality was mainly due to secondary metabolite toxicity, indicating that these fungi act as opportunistic entomopathogens. These findings highlight the potential of rhizosphere fungi as biological control agents for sustainable pest management.
SIGNIFICANCE STATEMENT
This study discovered entomopathogenic fungi from the chili (Capsicum annuum L.) rhizosphere that are capable of causing mortality in Spodoptera larvae, which can be beneficial for developing sustainable and eco-friendly pest management strategies. The findings provide insights into fungal-host interactions, and this study will help researchers to uncover the critical areas of rhizosphere-mediated biocontrol mechanisms that many researchers were not able to explore. Thus, a new theory on opportunistic entomopathogen activity in crop protection may be arrived at.
ACKNOWLEDGMENTS
The authors express their sincere gratitude to the Biology Laboratory, Faculty of Mathematics and Natural Sciences, Universitas Negeri Makassar, and the Plant and Horticultural Crop Protection Center (UPT BPTPH), Maros Regency, for providing facilities and technical support during this study.
REFERENCES
- Permadi, M.A., R.A. Lubis and I. Kinarang, 2019. Study of entomopathogenic fungi diversity from various rhizospheres of horticultural plants in Padangsidimpuan city [In Indonesian]. EKSAKTA: J. Penelitian Pembelajaran MIPA, 4: 1-8.
- Hidayat, T., K. Dinata, A. Ishak and E. Ramon, 2022. Identification of red chili pests and their control techniques in the Sari Mulyo farmer group, Sukasari Village, Air Periukan District, Seluma Regency, Bengkulu Province [In Indonesian]. J. Agrica Ekstensia, 16: 19-27.
- Suryani, A.I., H. Karim and A. Fudhail Majid, 2026. Effectiveness of plant-based nanoemulsion on immune response and morphology of Spodoptera frugiperda as an environmentally friendly pest control approach. Int. J. Agric. Biosci., 15: 871-878.
- Suryani, A.I., I.D. Daud, Melina and V.S. Dewi, 2024. Biological control of Spodoptera frugiperda pests with a combination of Mirabilis jalapa nanoemulsion and Beauveria bassiana. Asian J. Plant Sci., 23: 404-411.
- Muliani, Y., R. Robana and I. Mulyati, 2023. Application of the biological agent Trichoderma harzianum Rifai. to suppress Phytophtora infestans (Mont.) the cause of late blight disease in potato plants (Solanum tuberosum L.) [In Indonesian]. AGROScript: J. Appl. Agric. Sci., 5: 52-64.
- >Bamisile, B.S., K.S. Akutse, J.A. Siddiqui and Y. Xu, 2021. Model application of entomopathogenic fungi as alternatives to chemical pesticides: Prospects, challenges, and insights for next-generation sustainable agriculture. Front. Plant Sci., 12.
- Thalib, R., R. Fernando, Khodijah, D. Meidalima and S. Herlinda, 2013. Pathogeicity of Beauveria bassiana and Metarhizium anisopliae isolates from fresh swamp and tidal lowland, South Sumatra for Scirpophaga incertulas biological agents [In Indonesian]. J. Trop. Plant Pests Dis., 13: 10-18.
- Sharma, A., S. Sharma and P.K. Yadav, 2023. Entomopathogenic fungi and their relevance in sustainable agriculture: A review. Cogent Food Agric., 9.
- Abdullah, T., T. Kuswinanti, A. Nurariaty, I.D. Daud and A. Nasruddin et al., 2020. Application of Beauveria bassiana (Bals.) Vuil. (Hypocreales: Cordycipitaceae) in rice seed and its effect on mortality of green leaf hopper, Nephotettix virescens (Distant) (Homoptera: Cicadellidae). IOP Conf. Ser.: Earth Environ. Sci., 486.
- Prastowo, S., Soeharto, H.S. Addy and T. Handoyo, 2022. Virulence of Metarhizium isolated from infected Oryctes rhinoceros L. (Coleoptera: Scarabaeidae) larvae around coconut plantations in East Java, Indonesia. Egypt. J. Biol. Pest Control, 32.
- Oktariana, P., E. Rosa, W.A. Setiawan and N. Nurcahyani, 2024. The effectiveness of Nigrospora sp. and Penicillium sp. as entomopathogenic fungi against in Bactrocera sp. J. Exp. Biol. Biodivers., 11: 33-40.
- Tjampakasari, C.R., R. Agustini, I. Baihaki, S. Noor and A. Bustami, 2024. Slide culture as an indirect microscopic method for the identification of mold fungi [In Indonesian]. J. Sehat Indonesia, 6: 201-210.
- Barnett, H.L. and B.B. Hunter, 1972. Illustrated Genera of Imperfect Fungi. 3rd Edn., Burgess Publishing Company, Minneapolis, Minnesota, ISBN: 9780808702665, Pages: 241.
- Muljowati, J.S., O. Oedjijono, R.S. Dewi, A. Mariana and P. Chemeltorit, 2025. Exploration and morphological characterization of Trichoderma spp from organic waste at TPST Rempoah-Baturraden, Banyumas Regency. E3S Web of Conf., 609.
- Samson, R.A., J. Houbraken, U. Thrane, J.C. Frisvad and B. Andersen, 2010. Food and Indoor Fungi. CBS-KNAW Fungal Biodiversity Centre, Utrecht, Netherlands, ISBN: 9789070351823, Pages: 390.
- Watanabe, T., 2002. Pictorial Atlas of Soil and Seed Fungi: Morphologies of Cultured Fungi and Key to Species. 2nd Edn., CRC Press, Boca Raton, Florida, ISBN: 9780429075049, Pages: 504.
- Harman, G.E., C.R. Howell, A. Viterbo, I. Chet and M. Lorito, 2004. Trichoderma species-opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol., 2: 43-56.
- Samson, R.A., C.M. Visagie, J. Houbraken, S.B. Hong and V. Hubka et al., 2014. Phylogeny, identification and nomenclature of the genus Aspergillus. Stud. Mycol., 78: 141-173.
- Nyongesa, B.W., S. Okoth and V. Ayugi, 2015. Identification key for Aspergillus species isolated from maize and soil of Nandi County, Kenya. Adv. Microbiol., 5: 205-229.
- Jaber, L.R. and B.H. Ownley, 2018. Can we use entomopathogenic fungi as endophytes for dual biological control of insect pests and plant pathogens? Biol. Control, 116: 36-45.
- Fadhilah, F.R., A. Kafesa and S. Napita, 2021. Identification of Aspergillus sp mushrooms in wheat flour sold openly in Ciroyom market as raw materials for halal and mungle-free. Int. J. Mathla’ul Anwar Halal Issues, 1: 29-42.
- Anggraeni, D.N. and M. Usman, 2015. Test activity and identification of mushrooms on soil crop root rhizosphere banana (Musa paradisiaca) against Fusarium [In Indonesian]. BioLink: J. Biologi Lingkungan Industri Kesehatan, 1: 89-98.
- Kocsubé, S., G. Perrone, D. Magistà, J. Houbraken and J. Varga et al., 2016. Aspergillus is monophyletic: Evidence from multiple gene phylogenies and extrolites profiles. Stud. Mycol., 85: 199-213.
- Yusniwati, Nurbailis, Trizelia and M. Saragih, 2023. Potency of entomopathogen Beauveria bassiana fungus as biofertilizer and biostimulant to increase the plant growth of Cayenne pepper (Capsicum frutescens L.). IOP Conf. Ser.: Earth Environ. Sci., 1160.
- Trizelia, 2008. Pathogenicity of fungi entomopathogen Nomuraea rileyi (Farl.) Sams. against the pest Spodoptera exigua Hübner (Lepidoptera: Noctuidae) [In Tetum]. Indones. J. Entomol., 5: 108-115.
- Prayogo, Y., 2006. Distribution and efficacy of various entomopathogenic fungal genera against Riptortus linearis on soybean [In Indonesian]. J. Trop. Plant Pests Dis., 6: 14-22.
- Chouikhi, S., B.H. Assadi, K.G. Lebdi and M.S. Belkadhi, 2022. Efficacy of the entomopathogenic fungi Beauveria bassiana and Lecanicillium muscarium in the control of the tomato leaf miner, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Egypt. J. Biol. Pest Control, 32.
- Sun, B.D. and X.Z. Liu, 2008. Occurrence and diversity of insect-associated fungi in natural soils in China. Appl. Soil Ecol., 39: 100-108.
- Sun, B.D., H.Y. Yu, A.J. Chen and X.Z. Liu, 2008. Insect-associated fungi in soils of field crops and orchards. Crop Prot., 27: 1421-1426.
How to Cite this paper?
APA-7 Style
Mutia,
D., Rachmawaty, Suryani,
A.I. (2026). Isolation and Identification of Entomopathogenic Fungi from Chili Rhizosphere Against Fall Armyworm. Trends in Biological Sciences, 2(2), 189-200. https://doi.org/10.21124/tbs.2026.189.200
ACS Style
Mutia,
D.; Rachmawaty; Suryani,
A.I. Isolation and Identification of Entomopathogenic Fungi from Chili Rhizosphere Against Fall Armyworm. Trends Biol. Sci 2026, 2, 189-200. https://doi.org/10.21124/tbs.2026.189.200
AMA Style
Mutia
D, Rachmawaty, Suryani
AI. Isolation and Identification of Entomopathogenic Fungi from Chili Rhizosphere Against Fall Armyworm. Trends in Biological Sciences. 2026; 2(2): 189-200. https://doi.org/10.21124/tbs.2026.189.200
Chicago/Turabian Style
Mutia, Dinda, A. Irma Suryani.
2026. "Isolation and Identification of Entomopathogenic Fungi from Chili Rhizosphere Against Fall Armyworm" Trends in Biological Sciences 2, no. 2: 189-200. https://doi.org/10.21124/tbs.2026.189.200

This work is licensed under a Creative Commons Attribution 4.0 International License.




