Prevalence of Intestinal Parasites and Plasmodium Co-Infection in Two Wards of Hong Local Government Area, Adamawa State, Nigeria
| Received 26 Sep, 2025 |
Accepted 26 Mar, 2026 |
Published 30 Jun, 2026 |
Background and Objective: Co-infection with soil-transmitted helminths (STHs) and Plasmodium falciparum poses significant health risks, including anaemia and increased mortality. This study aimed to determine the prevalence of STHs and P. falciparum among nomadic settlements in two selected wards of Hong Local Government Area, Adamawa State, Nigeria. Materials and Methods: A prospective cross-sectional study was conducted in Daksiri and Thilband wards. Stool and blood samples were collected from 200 participants and screened for intestinal parasites and P. falciparum using standard laboratory techniques. Structured questionnaires assessed participants’ knowledge and practices regarding intestinal parasites and malaria. Data were analyzed using SPSS version 36.0. Results: Of the 200 participants, 35.5% were infected with at least one intestinal parasite, while 25.5% tested positive for P. falciparum. Entamoeba histolytica was the most prevalent intestinal parasite (7.6%), and Strongyloides stercoralis was the least common (1.2%). The 11-20 years age group had the highest prevalence for both intestinal parasites (14%) and P. falciparum (7%), whereas the ≥41 years age group had the lowest prevalence (intestinal parasites: 3.5%; P. falciparum: 1.5%). Males exhibited higher infection rates than females for both intestinal parasites (40% vs. 35%) and P. falciparum (28.4% vs. 22.4%), with statistical significance observed for intestinal parasites (p<0.05). Geographically, Thilbang ward recorded the highest intestinal parasite prevalence (41%), while Daksiri ward had the highest P. falciparum prevalence (31%). Knowledge and practices varied: 69% of participants were aware of intestinal worms, 75% used well water, 93% used pit latrines, and 83.5% reported using insecticide-treated nets. Conclusion: The high prevalence of STHs and P. falciparum co-infection highlights the need for targeted interventions, including health education, improved sanitation, and treatment of infected individuals, to reduce associated morbidity and prevent complications.
| Copyright © 2026 Innocent et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Malaria and intestinal parasitic infections are both known to cause severe haematological abnormalities, including anaemia, thrombocytopenia, and impaired immune responses. These infections remain major contributors to morbidity and mortality in Africa, particularly in resource-limited tropical and subtropical regions such as Sub-Saharan Africa1. Although Nigeria has implemented several preventive and control strategies-such as vector control, mass drug administration, and improved access to chemotherapy-the complete elimination and eradication of malaria and intestinal parasites remain challenging. This is largely due to persistent problems such as poor sanitation, inadequate sewage systems, improper waste disposal, and widespread mosquito breeding environments, which continue to pose serious public health threats across the country2.
Globally, malaria remains one of the leading causes of death in developing countries. According to recent estimates, over 3.3 billion people are still at risk of malaria worldwide3. In 2022 alone, approximately 249 million malaria cases and 608,000 deaths were recorded globally, with about 94% of these deaths occurring in Sub-Saharan Africa3. Children under five years, pregnant women, and immunocompromised individuals-including people living with HIV/AIDS-remain the most vulnerable groups. Africa continues to bear the heaviest burden of malaria, accounting for about 93% of global cases and 96% of global deaths, with Plasmodium falciparum responsible for over 99% of malaria infections in Nigeria and most of West Africa2,3.
Intestinal parasitic infections (IPIs) also represent a major public health burden, particularly in low-income communities of Sub-Saharan Africa. These infections thrive in areas characterized by warm and humid climates, poor personal hygiene, unsafe water supply, and inadequate sanitation facilities3,4. The parasites responsible for IPIs include protozoans such as Cryptosporidium species, Cyclospora cayetanensis, Isospora belli, Microsporidia species, and Entamoeba histolytica, as well as helminths including Ascaris lumbricoides, Trichuris trichiura, hookworms (Ancylostoma duodenale and Necator americanus), Schistosoma mansoni, and Taenia species5.
Soil-transmitted helminths (STHs) are intestinal roundworms transmitted through soil contaminated with human faeces. The major STHs of public health importance include Ascaris lumbricoides (roundworm), Trichuris trichiura (whipworm), and the hookworms (Ancylostoma duodenale and Necator americanus). More than 80% of the global burden of STH infections occurs in developing countries, particularly in Sub-Saharan Africa, Asia, and Latin America5,6.
Intestinal helminth infections remain among the most widespread infections in the developing world, especially in communities with limited access to safe water and sanitary facilities6. Globally, over 1.5 billion people-nearly 24% of the world’s population-are infected with soil-transmitted helminths. Ascaris lumbricoides, Trichuris trichiura, and hookworms infect an estimated 898 million, 460 million, and 470 million people worldwide, respectively6,7. These infections lead to impaired digestion and nutrient absorption, chronic blood loss, reduced physical fitness, loss of appetite, poor cognitive development, and decreased school performance in children. In adults, helminth infections are associated with reduced work productivity, economic loss, and impaired physical capacity8,9.
Ascaris lumbricoides, the causative agent of ascariasis, is one of the most prevalent neglected tropical diseases worldwide. It is estimated that nearly one-sixth of the global population has been infected with Ascaris at some point in life6. Although most infections are asymptomatic, heavy worm burdens may result in intestinal obstruction, malnutrition, impaired growth, pulmonary complications, and iron-deficiency anaemia-especially among children6.
Trichuris trichiura, commonly known as whipworm, causes trichuriasis, an intestinal infection of the large bowel. Transmission is closely associated with poor hygiene, contaminated food and water, and ingestion of eggs from moist, shaded soil polluted with human faeces7. Chronic trichuriasis may lead to abdominal pain, diarrhoea, rectal prolapse, growth retardation, and impaired cognitive development in children9. This study determines the prevalence and distribution of intestinal parasitic infections (IPIs) caused by protozoans and helminths among the study population.
MATERIALS AND METHODS
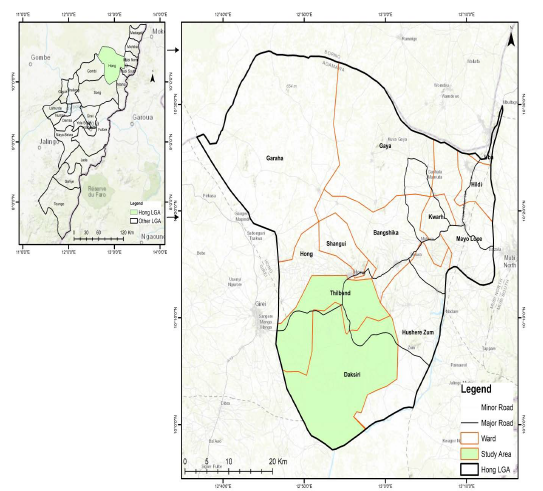
Study area: Hong Local Government Area is located in the Adamawa Central Senatorial zone of Adamawa State, North-East Nigeria (Fig. 1). It lies between Latitude 10°13'543N and Longitude 12°55'493E of the Greenwich meridian. It is bounded to the South by Song Local Government, to the South East by Maiha Local Government, Borno State in the North, Mubi Local Government to the East, and Gombi Local Government to the West. It is located between the rivers Killanyi and Hawul. It occupies a landmass of about 2,626 km2 which is about 2.9% of the Adamawa State landmass. It has an estimated population of 169,126 based on the 2006 census, made up of 83,805 males and 85,321 females. Given population density of 86.10 m2. majority of the people in the area are farmers, and business and civil services are also part of their occupation (National Population Commission (NPC, 2006). It has 12 political wards that are subdivided into two State Constituencies, with each having six wards. Hong Constituency comprises Bangshika ward, Daksiri ward, Hong ward, Husherizum ward, Shangui ward and Thilbang ward. Uba/Gaya Constituency comprises Garaha ward, Gaya ward, Hildi ward, Kwarhi ward, Mayo-lope ward, and Uba ward. The Local Government lies within the tropical region having dry and rainy seasons. The rainy season begins from April and lasts until October with a mean annual rainfall of 700mm. the wettest months are August and September. The dry season starts from November and ends in March, which is the hammatan period when dust-laden North East trade winds blow from the Sahara Desert with a marked effect on the climate of the area. Temperature varies from a minimum of 23°C to 36°C10. The crops cultivated in this area are groundnuts, Guinea corn, maize, rice, beans, and so on (Fig. 1).
Hong is largely covered by short-grass savanna. The LGA has a lot of educated elites. It has over 50 primary health care facilities, 1 Federal Medical Centre and state College of Education. The Kilba people (also called Həba) are the major ethnic group in the Hong Local Government Area of Adamawa State, followed by Marghi and Settlers such as Hausa, Fulani, Michika etc.
Ethical consent/clearance: Approval and permission for this study were granted by the Adamawa State Ministry of Health Ethical Committee on Health Research. An introductory letter was obtained from the Department of Zoology, Modibbo Adama University, Yola. The selected communities were informed about the study, and their permission was sought and obtained. Written and verbal consent was also obtained from patients/subjects for voluntary participation.
Study design and population: A descriptive cross-sectional community-based study was carried out from March to August 2022 in Thilband and Daksiri wards. The two communities serve the population of the nomads and their environs. Both stool and blood samples were collected from each subject. The stool samples were used for screening of intestinal parasites, while blood samples from the patients were used for the screening of malaria parasites. The study involves nomadic individuals within the age of 18-60 years who reside in Daksiri and Thilband wards in the Hong Local Government.
|
A total of two hundred (200) nomad subjects from both wards were selected for this study using the Yamane Taro formula:11
Where:
| n | = | Sample size | |
| N | = | Population size | |
| e | = | Margin of error (level of precision, expressed as a decimal, e.g., 5% = 0.05) |
A questionnaire was also distributed to respondents for data collection.
Parasitological and malaria detection methods
Sample collection: A total of 200 participants from Daksiri and Thilband wards of Hong LGA, Adamawa State, Nigeria, were enrolled in the study. Stool and blood samples were collected to determine intestinal parasite and Plasmodium falciparum prevalence, respectively.
Stool samples: Participants provided fresh stool specimens in wide-mouthed, sterilized, leak-proof containers labeled with unique identification numbers. Each participant also completed a structured questionnaire to provide socio-demographic information (age, gender, occupation, educational level, location, and healthcare facility visited) and to assess their knowledge on intestinal parasites and malaria. Participants were advised to observe basic hygiene and protective measures, such as wearing footwear, to prevent deterioration of trophozoites and cysts.
Blood samples: Finger-prick blood samples were collected using sterile lancets. Approximately 2-3 drops of blood were used to prepare thick and thin smears on clean, labeled microscope slides. The slides were air-dried at room temperature and stored appropriately before staining.
Macroscopic examination of stool samples: Stool samples were examined macroscopically for colour, consistency, odour, presence of mucus, blood, bloody mucus, visible adult worms, or segments of intestinal helminths in accordance with current World Health Organization laboratory diagnostic guidelines12.
Microscopic examination of stool samples
Saline wet mount technique: A small portion of each stool sample was emulsified in a drop of 0.85-0.9% physiological saline on a clean glass slide, covered with a coverslip, and examined under ×10 and ×40 objective lenses for the detection of ova, larvae, cysts, and motile trophozoites. This procedure was conducted following updated standard parasitological diagnostic methods13.
Formol-ether (ethyl acetate) concentration technique: Approximately 1-2 g (walnut-sized portion) of stool was emulsified in 10 mL of normal saline and filtered through fine gauze into a centrifuge tube. The filtrate was centrifuged at 500-600 g for 10 minutes. The supernatant was discarded, and the sediment was fixed with 10% formalin. Two millilitres of ethyl acetate were added and the mixture centrifuged at 450 g for 10 minutes, producing four distinct layers. The top three layers were decanted, and the sediment was examined microscopically under ×10 and ×40 objectives for parasitic ova, larvae, and cysts. Identification was based on morphological features using standard parasitology identification keys12.
Blood smear examination for malaria: Blood smears were stained with 10% Giemsa solution for 10-15 minutes, rinsed, and air-dried following WHO protocols14. Thick smears were used to detect the presence of Plasmodium parasites, and thin smears were used for species identification based on morphological characteristics. Parasite density was estimated by counting the number of asexual parasites per 200 white blood cells in thick smears, assuming a standard WBC count of 8,000/μL. All slides were read independently by two microscopists, with discrepancies resolved by a third expert microscopist to ensure quality control.
Data analysis: Data were analyzed using SPSS version 36.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics summarized socio-demographic characteristics, prevalence of intestinal parasites, and Plasmodium falciparum infection as frequency and percentage (%). Associations between categorical variables, including infection status and participants’ age, gender, and location, were determined using Chi-square (χ2) tests. A p-value<0.05 was considered statistically significant.
RESULTS
The prevalence of intestinal parasites and Plasmodium falciparum co-infection among participants in Daksiri and Thilband wards of Hong LGA is summarized in Table 1-3. Table 1 shows the overall prevalence of intestinal parasites, where 75 (37.5%) of the 200 samples were positive for intestinal parasites only. Seven parasite species were identified, with Entamoeba histolytica being the most prevalent 16 (8.0%), followed by Giardia lamblia 13 (6.5%), while Strongyloides stercoralis was the least common 4 (2.0%). All positive cases were single infections. Co-infection with intestinal parasites and P. falciparum was observed in 51 (25.5%) of participants. Table 2 presents prevalence in relation to age. The highest intestinal parasite prevalence was recorded among participants aged 11-20 years, 28 (14.0%), followed by the 1-10 years’ age group, 16 (8.0%), and the lowest prevalence was among participants aged 41 years and above, 7 (3.5%), with a statistically significant difference (p<0.05). P. falciparum prevalence was also highest in the 11-20 years age group, 14 (7.0%), and lowest in the 41 years and above group, 3 (1.5%). Table 3 shows prevalence in relation to gender. Males exhibited higher infection rates than females for both intestinal parasites, 41 (40.0%) vs. 34 (35.0%), and P. falciparum, 29 (28.4%) vs. 22 (22.4%), with statistically significant differences (p<0.05). These findings indicate that adolescents and male participants are disproportionately affected by intestinal parasite infections and P. falciparum co-infection in the study area, highlighting the need for targeted interventions.
| Table 1: | Overall prevalence of intestinal parasites and Plasmodium co-infection in the study area | |||
| Parasite species no exam = 200 | Occurance | Occurance (%) |
| Entamoeba histolytica | 16 | 8 |
| Gardia lamblia | 13 | 6.5 |
| Ascaris lumbricoides | 11 | 5.5 |
| Hymenolepis nana | 8 | 4 |
| Ancylostoma duodenale | 14 | 7 |
| Taenia saginata | 9 | 4.5 |
| Strongyloides stercoralis | 4 | 2 |
| Total | 75 | 37.5 |
| Co-infection (Intestinal parasite+P. falciparum) | 51 | 25.5 |
| Table 2: | Prevalence of intestinal parasites and Plasmodium co-infection in relation to age in Daksiri and Thilband Ward, Hong LGA | |||
| Age range (Yrs) | No. examined (Intestinal parasites) |
No. positive | Prevalence (%) | No. examined (Plasmodium co-infection) |
No. positive | Prevalence (%) |
| 01-Oct | 46 | 16 | 8 | 46 | 11 | 5.5 |
| Nov-20 | 63 | 28 | 14 | 63 | 14 | 7 |
| 21-30 | 38 | 14 | 7 | 38 | 10 | 5 |
| 31-40 | 32 | 10 | 5 | 32 | 13 | 6.5 |
| 41≥ | 21 | 7 | 3.5.0 | 21 | 3 | 1.5 |
| Total | 200 | 75 | 37.5 | 200 | 51 | 25.5 |
| Table 3: | Prevalence of intestinal parasites and Plasmodium co-infection in relation to gender in Daksiri and Thilband Ward, Hong LGA | |||
| Gender | No. examined (Intestinal parasites) |
No. positive | Prevalence (%) | No. examined (Plasmodium co-infection) |
No. positive | Prevalence (%) |
| Male | 102 | 41 | 40 | 102 | 29 | 28.4 |
| Female | 98 | 34 | 35 | 98 | 22 | 22.4 |
| Total | 200 | 75 | 37.5 | 200 | 51 | 25.5 |
| Table 4: | Prevalence of Intestinal Parasites and Plasmodium co-infection in relation to location in Hong LGA | |||
| Ward | No. examined (Intestinal parasites) |
No. positive | Prevalence (%) | No. examined (Plasmodium co-infection) |
No. positive | Prevalence (%) |
| Daksiri | 100 | 41 | 41 | 100 | 20 | 20 |
| Thilband | 100 | 34 | 34 | 100 | 31 | 31 |
| Total | 200 | 75 | 37.5 | 200 | 51 | 25.5 |
The prevalence of intestinal parasites and Plasmodium falciparum in relation to location and the knowledge of study participants is summarized in Table 4 and 5. Out of 200 samples examined, 100 (50.0%) were from Daksiri ward and 100 (50.0%) from Thilband ward. Daksiri ward recorded the highest intestinal parasite prevalence 41 (41.0%), while Thilband ward had the highest P. falciparum prevalence, 31 (31.0%). Conversely, Thilband ward had the lowest intestinal parasite prevalence, 34 (34.0%), and Daksiri ward had the lowest malaria prevalence 20 (20.0%). Chi-square analysis showed no statistically significant difference in the prevalence of co-infection by location (p>0.05).
| Table 5: | Knowledge on intestinal parasites and Plasmodium co-infection among study subjects | |||
| Variable n = 200 | Frequency | Percentages (%) |
| Knowledge of intestinal parasite | ||
| Yes | 138 | 69 |
| No | 62 | 31 |
| Deworming | ||
| Last month | 11 | 5.5 |
| Last three months | 23 | 11.5 |
| Don’t know | 166 | 83 |
| Water source | ||
| Borehole | 42 | 22.5 |
| Well | 150 | 75 |
| Stream | 8 | 4 |
| Toilet facility in your area | ||
| Pit | 186 | 93 |
| Water closet | 0 | 0 |
| Bush | 14 | 7 |
| Hand washing after going to the toilet | ||
| Yes | 123 | 61.5 |
| No | 38 | 19 |
| Not always | 39 | 19.5 |
| Fruits and vegetables washing before consumption | ||
| Yes | 46 | 23 |
| No | 15 | 7.5 |
| Not always | 139 | 69.5 |
| Knowledge of malaria | ||
| Yes | 156 | 78 |
| No | 44 | 22 |
| Last time malaria was treated | ||
| Last month | 43 | 21.5 |
| 3 months ago | 129 | 64.5 |
| Didn’t know | 28 | 14 |
| Use of treated mosquito net | ||
| Yes | 167 | 83.5 |
| No | 33 | 16.5 |
The level of knowledge on intestinal parasites and malaria among participants is presented in Table 5. Overall, 138 (69.0%) of participants had heard of intestinal worms, whereas 62 (31.0%) had not. Most participants were unaware of their last deworming, 166 (83.0%), while 11 (5.5%) reported deworming in the past month. Well water was the main source of drinking water for 150 (75.0%) participants, while 8 (4.0%) used streams. Pit latrines were the predominant toilet facility 186 (93.0%), while 14 (7.0%) practiced open defecation. Hand hygiene practices showed that 123 (61.5%) washed their hands after defecation, whereas 38 (19.0%) did not. Regarding food hygiene, 139 (69.5%) did not consistently wash fruits and vegetables before consumption. Awareness of malaria was high, with 156 (78.0%) having heard of the disease, while 44 (22.0%) were unaware. Malaria treatment in the last three months was reported by 129 (64.5%) participants, while 28 (14.0%) were unsure. Regular use of insecticide-treated nets (ITNs) was reported by 167 (83.5%) participants, whereas 33 (16.5%) did not use ITNs.
DISCUSSION
This study investigated the burden of intestinal parasites and Plasmodium falciparum co-infection among nomadic populations in two wards of Hong Local Government Area, Adamawa State, Nigeria. Out of the 200 participants examined, 37.5% were infected with at least one intestinal parasite, while 26.0% had co-infection with Plasmodium falciparum. The intestinal parasites identified-Entamoeba histolytica, Giardia lamblia, Ancylostoma duodenale, Taenia saginata, Hymenolepis nana, Ascaris lumbricoides, and Strongyloides stercoralis-are consistent with parasites widely reported across Nigeria and other Sub-Saharan African countries3,4.
Entamoeba histolytica emerged as the most prevalent parasite, aligning with recent community-based studies in Northern Nigeria that highlight its dominance in areas with limited access to potable water and sanitation facilities14. Giardia lamblia ranked second in prevalence, supporting findings from both Nigerian and global studies that associate giardiasis with poor water quality and overcrowded living conditions15,16. Strongyloides stercoralis recorded the lowest prevalence, which agrees with recent studies that attribute its reduced detection to underdiagnosis and the need for specialized laboratory methods16,17.
The malaria prevalence observed in this study is also comparable with reports from malaria-endemic regions in Nigeria, particularly during the rainy season when mosquito breeding intensifies2,18.
Age-specific analysis revealed that adolescents and young adults were more vulnerable to both intestinal parasites and malaria. This trend mirrors recent rural studies which attribute higher infection rates in this age group to increased outdoor activities, farming, herding, and poor adherence to preventive measures5,19. Older adults showed relatively lower infection rates, possibly due to accumulated immunity, better hygiene awareness, and repeated treatment exposure, as documented in similar endemic communities20.
Gender-related differences showed a higher prevalence of infection among males compared to females. This observation agrees with recent Nigerian and African studies linking male dominance in outdoor occupations such as farming, grazing, and fishing to increased exposure to both malaria vectors and contaminated environments3,21. Females recorded lower prevalence, which may be associated with better hygiene practices, earlier health-seeking behavior, and possible immunological advantages21,22.
Marked spatial variations were observed between the two wards studied. The higher prevalence of intestinal parasites in one ward is likely associated with poor environmental sanitation, unsafe water supply, open defecation, and frequent barefoot walking. Similar environmental risk factors have been widely reported in rural Nigerian studies4,14. Conversely, higher malaria prevalence in the other ward may reflect differences in ecological conditions, vector density, housing structures, and availability of insecticide-treated nets, as documented in other endemic communities22,23.
Significant gaps were observed in knowledge and preventive practices, including irregular deworming, reliance on unsafe water sources, poor hand hygiene, and inconsistent use of mosquito nets. These findings are consistent with recent reports indicating that low literacy levels, cultural practices, and limited access to health education continue to drive persistent parasitic transmission among nomadic and rural populations2,12. These results strongly highlight the urgent need for integrated interventions involving health education, improved sanitation, routine deworming, and sustained malaria vector control strategies tailored to nomadic communities.
CONCLUSION
This study demonstrates a substantial prevalence of intestinal parasites and Plasmodium falciparum co-infection among nomadic populations in Hong LGA, Adamawa State, Nigeria, with adolescents, young adults, and males being the most affected groups. The observed patterns suggest that inadequate sanitation, poor hygiene practices, limited awareness, and occupational or behavioral exposure contribute significantly to the persistence and transmission of these infections.
To address these public health challenges, targeted interventions are recommended. These include comprehensive community health education programs on hygiene, safe water usage, and malaria prevention; implementation of regular deworming campaigns combined with malaria control measures such as insecticide-treated nets; improvements in environmental sanitation and access to potable water; and promotion of early health-seeking behavior across all age groups, particularly among males and adolescents. These strategies will help reduce the burden of intestinal parasites and malaria co-infections and improve overall health outcomes in these communities.
SIGNIFICANCE STATEMENT
This study provides crucial insights into the burden of co-infection with soil-transmitted helminths and Plasmodium falciparum among nomadic communities in Adamawa State, Nigeria. By identifying age, gender, and geographic patterns of infection, as well as knowledge and hygiene practices, the findings underscore the urgent need for integrated control strategies. Targeted health education, improved sanitation, and timely treatment can reduce morbidity, prevent severe complications, and support overall community health.
REFERENCES
- Semakula, H.M., S. Liang, P.I. Mukwaya, F. Mugagga and M. Swahn et al., 2023. Determinants of malaria infections among children in refugee settlements in Uganda during 2018-2019. Infect. Dis. Poverty, 12.
- WHO, 2023. World Malaria Report 2023. WHO, Geneva, ISBN: 978-92-4-008617-3, Pages: 356.
- Tyoalumun, K., S. Abubakar and N. Christopher, 2016. Prevalence of intestinal parasitic infections and their association with nutritional status of rural and urban pre-school children in Benue State, Nigeria. Int. J. Maternal Child Health AIDS, 5: 146-152.
- Amisu, B.O., O.J. Okesanya, N.O. Olaleke, C.O. Ologun and D.E. Lucero-Prisno III et al., 2023. Socio-environmental determinants of parasitic intestinal infections among children: A cross-sectional study in Nigeria. J. Global Health Sci., 5.
- WHO, 2023. Soil-Transmitted Helminth Infections. World Health Organization, Geneva, Switzerland.
- Jourdan, P.M., P.H.L. Lamberton, A. Fenwick and D.G. Addiss, 2018. Soil-transmitted helminth infections. Lancet, 391: 252-265.
- Chen, J., Y. Gong, Q. Chen, S. Li and Y. Zhou, 2024. Global burden of soil-transmitted helminth infections, 1990-2021. Infect. Dis. Poverty, 13.
- Turner, H.C., J.E. Truscott, A.A. Bettis, K.V. Shuford and J.C. Dunn et al., 2015. An economic evaluation of expanding hookworm control strategies to target the whole community. Parasites Vectors, 8.
- Pabalan, N., E. Singian, L. Tabangay, H. Jarjanazi, M.J. Boivin and A.E. Ezeamama, 2018. Soil-transmitted helminth infection, loss of education and cognitive impairment in school-aged children: A systematic review and meta-analysis. PLoS Negl. Trop. Dis., 12.
- Adelekan, I.O., 2010. Vulnerability of poor urban coastal communities to flooding in Lagos, Nigeria. Environ. Urbanization, 22: 433-450.
- Yamane, T., 1967. Statistics: An Introductory Analysis. 2nd Edn., Harper and Row, New York, Pages: 919.
- WHO, 2019. Bench Aids for the Diagnosis of Intestinal Parasites. 2nd Edn., World Health Organization, Geneva, Switzerland, ISBN: 978-92-4-151534-4, Pages: 32.
- WHO, 2009. Malaria Microscopy Quality Assurance Manual: V.1. WHO Regional Office for the Western Pacific, Geneva, Switzerland, 9789290614227, Pages: 125.
- Isma’l, I., A. Ahmed, K. Abdullahi, A.M. Yusuf, Z.A. Yar’adua and S.M. Gidado, 2024. Prevalence and risk factors of intestinal parasitic infections among schoolchildren in Katsina Metropolis, Katsina State, Nigeria. Sahel J. Life Sci. FUDMA, 2: 62-68.
- Tijani, M.K., P.C. Köster, I. Guadano-Procesi, I.S. George and E. Abodunrin et al., 2023. High diversity of Giardia duodenalis assemblages and sub-assemblages in asymptomatic school children in Ibadan, Nigeria. Trop. Med. Infect. Dis., 8.
- WHO, 2018. Guidelines on Sanitation and Health. World Health Organization, Rome, Italy, ISBN: 978-92-4-151470-5, Pages: 220.
- Buonfrate, D., D. Bisanzio, G. Giorli, P. Odermatt and T. Fürst et al., 2020. The global prevalence of Strongyloides stercoralis infection. Pathogens, 9.
- Schär, F., U. Trostdorf, F. Giardina, V. Khieu and S. Muth et al., 2013. Strongyloides stercoralis: Global distribution and risk factors. PLoS Negl. Trop. Dis., 7.
- Awan, A. and T. Alcindor, 2019. Secondary polycythemia in a sarcoma patient: A commentary about cediranib. Curr. Oncol., 26: e1-e2.
- Pullan, R.L. and S.J. Brooker, 2012. The global limits and population at risk of soil-transmitted helminth infections in 2010. Parasites Vectors, 5.
- Mukhtar, A., M. Ismail, A. Usman, I.A. Odetokun and M.A. Ibrahim et al., 2026. Prevalence and associated risk factors of asymptomatic malaria in Nigeria: A systematic review and meta-analysis. Malar. J., 25.
- Klein, S.L. and K.L. Flanagan, 2016. Sex differences in immune responses. Nat. Rev. Immunol., 16: 626-638.
- National Malaria Elimination Programme, 2021. National Malaria Strategic Plan (NMSP) of Nigeria: 2021-2025. Federal Ministry of Health, Abuja, Nigeria, Pages: 106.
How to Cite this paper?
APA-7 Style
Innocent,
A., Qadeer,
M.A., Atinga,
A., Benson,
O. (2026). Prevalence of Intestinal Parasites and Plasmodium Co-Infection in Two Wards of Hong Local Government Area, Adamawa State, Nigeria. Trends in Biological Sciences, 2(2), 179-188. https://doi.org/10.21124/tbs.2026.179.188
ACS Style
Innocent,
A.; Qadeer,
M.A.; Atinga,
A.; Benson,
O. Prevalence of Intestinal Parasites and Plasmodium Co-Infection in Two Wards of Hong Local Government Area, Adamawa State, Nigeria. Trends Biol. Sci 2026, 2, 179-188. https://doi.org/10.21124/tbs.2026.179.188
AMA Style
Innocent
A, Qadeer
MA, Atinga
A, Benson
O. Prevalence of Intestinal Parasites and Plasmodium Co-Infection in Two Wards of Hong Local Government Area, Adamawa State, Nigeria. Trends in Biological Sciences. 2026; 2(2): 179-188. https://doi.org/10.21124/tbs.2026.179.188
Chicago/Turabian Style
Innocent, A., M. A. Qadeer, A. Atinga, and O. Benson.
2026. "Prevalence of Intestinal Parasites and Plasmodium Co-Infection in Two Wards of Hong Local Government Area, Adamawa State, Nigeria" Trends in Biological Sciences 2, no. 2: 179-188. https://doi.org/10.21124/tbs.2026.179.188

This work is licensed under a Creative Commons Attribution 4.0 International License.




